QUESTION IMAGE
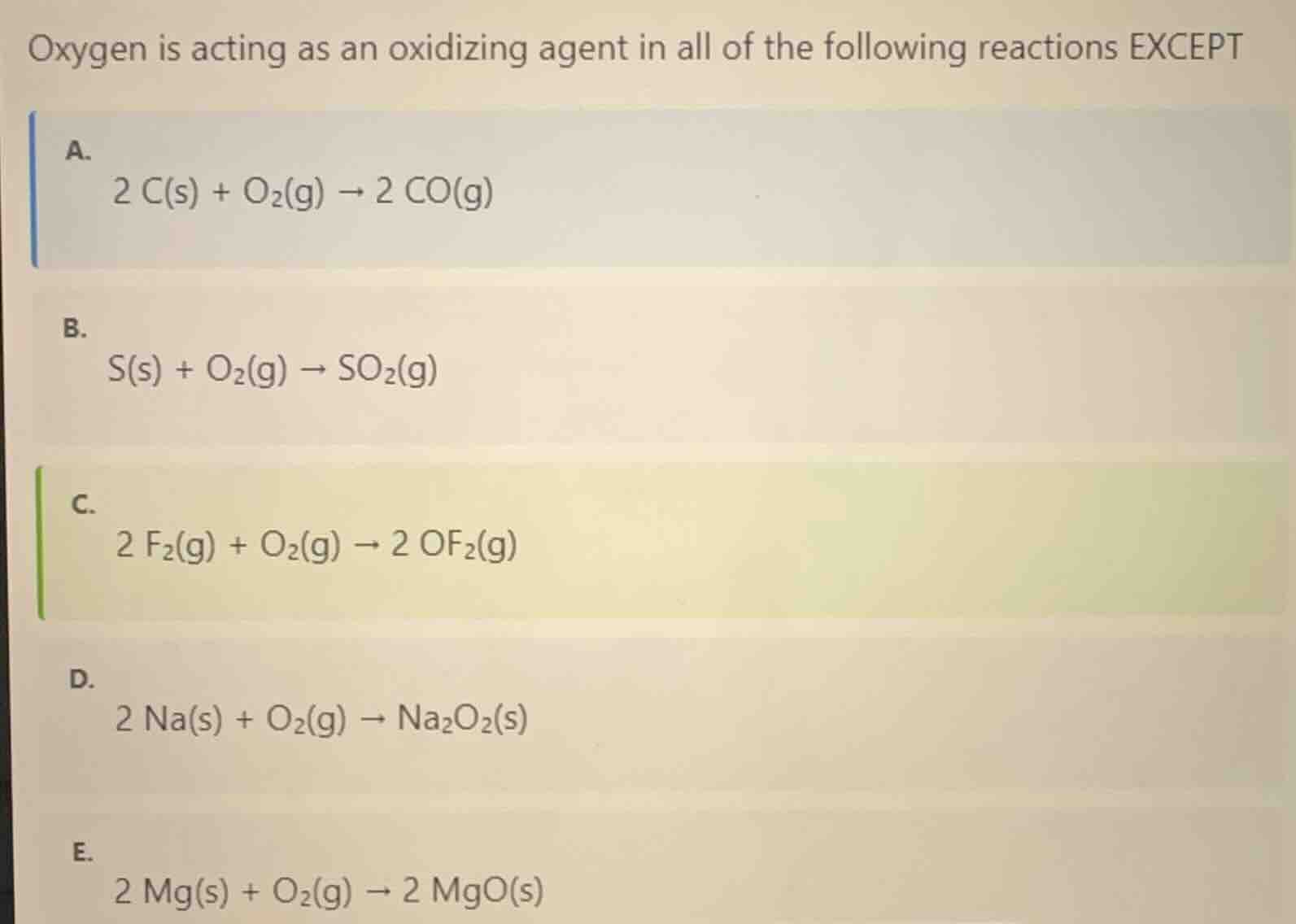
Question
oxygen is acting as an oxidizing agent in all of the following reactions excepta.\\(\ce{2 c(s) + o2(g) -> 2 co(g)}\\)b.\\(\ce{s(s) + o2(g) -> so2(g)}\\)c.\\(\ce{2 f2(g) + o2(g) -> 2 of2(g)}\\)d.\\(\ce{2 na(s) + o2(g) -> na2o2(s)}\\)e.\\(\ce{2 mg(s) + o2(g) -> 2 mgo(s)}\\)
Brief Explanations
To determine when oxygen is not an oxidizing agent, we analyze the oxidation states:
- In an oxidizing agent, the element is reduced (oxidation state decreases).
- For option A: C goes from 0 to +2 (oxidized), O in \(O_2\) (0) goes to -2 (reduced) → \(O_2\) is oxidizing agent.
- For option B: S goes from 0 to +4 (oxidized), O in \(O_2\) (0) goes to -2 (reduced) → \(O_2\) is oxidizing agent.
- For option C: F in \(F_2\) (0) goes to -1 (reduced), O in \(O_2\) (0) goes to +2 (oxidized) → \(O_2\) is oxidized (acting as reducing agent), not oxidizing agent.
- For option D: Na goes from 0 to +1 (oxidized), O in \(O_2\) (0) goes to -1 (reduced) → \(O_2\) is oxidizing agent.
- For option E: Mg goes from 0 to +2 (oxidized), O in \(O_2\) (0) goes to -2 (reduced) → \(O_2\) is oxidizing agent.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. \( 2 F_2(g) + O_2(g)
ightarrow 2 OF_2(g) \)