QUESTION IMAGE
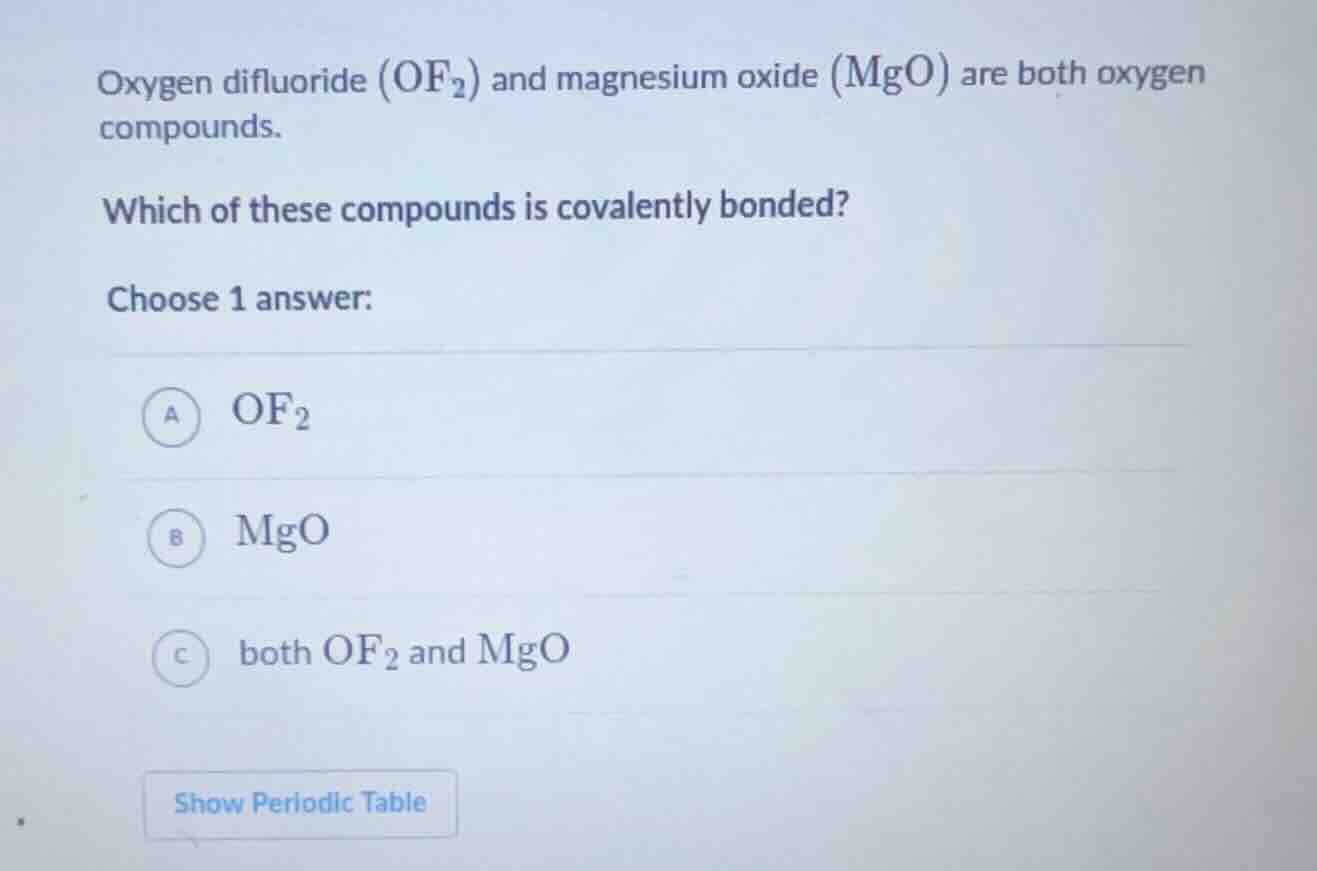
Question
oxygen difluoride (\\(\ce{of2}\\)) and magnesium oxide (\\(\ce{mgo}\\)) are both oxygen compounds.
which of these compounds is covalently bonded?
choose 1 answer:
\\(\boldsymbol{\text{a}}\\) \\(\ce{of2}\\)
\\(\boldsymbol{\text{b}}\\) \\(\ce{mgo}\\)
\\(\boldsymbol{\text{c}}\\) both \\(\ce{of2}\\) and \\(\ce{mgo}\\)
To determine covalent bonding, we check the types of elements. Covalent bonds form between nonmetals, while ionic bonds form between metals and nonmetals. Oxygen (O) and fluorine (F) are both nonmetals, so \( \text{OF}_2 \) has covalent bonds. Magnesium (Mg) is a metal and oxygen (O) is a nonmetal, so \( \text{MgO} \) has ionic bonds.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \( \text{OF}_2 \)