QUESTION IMAGE
Question
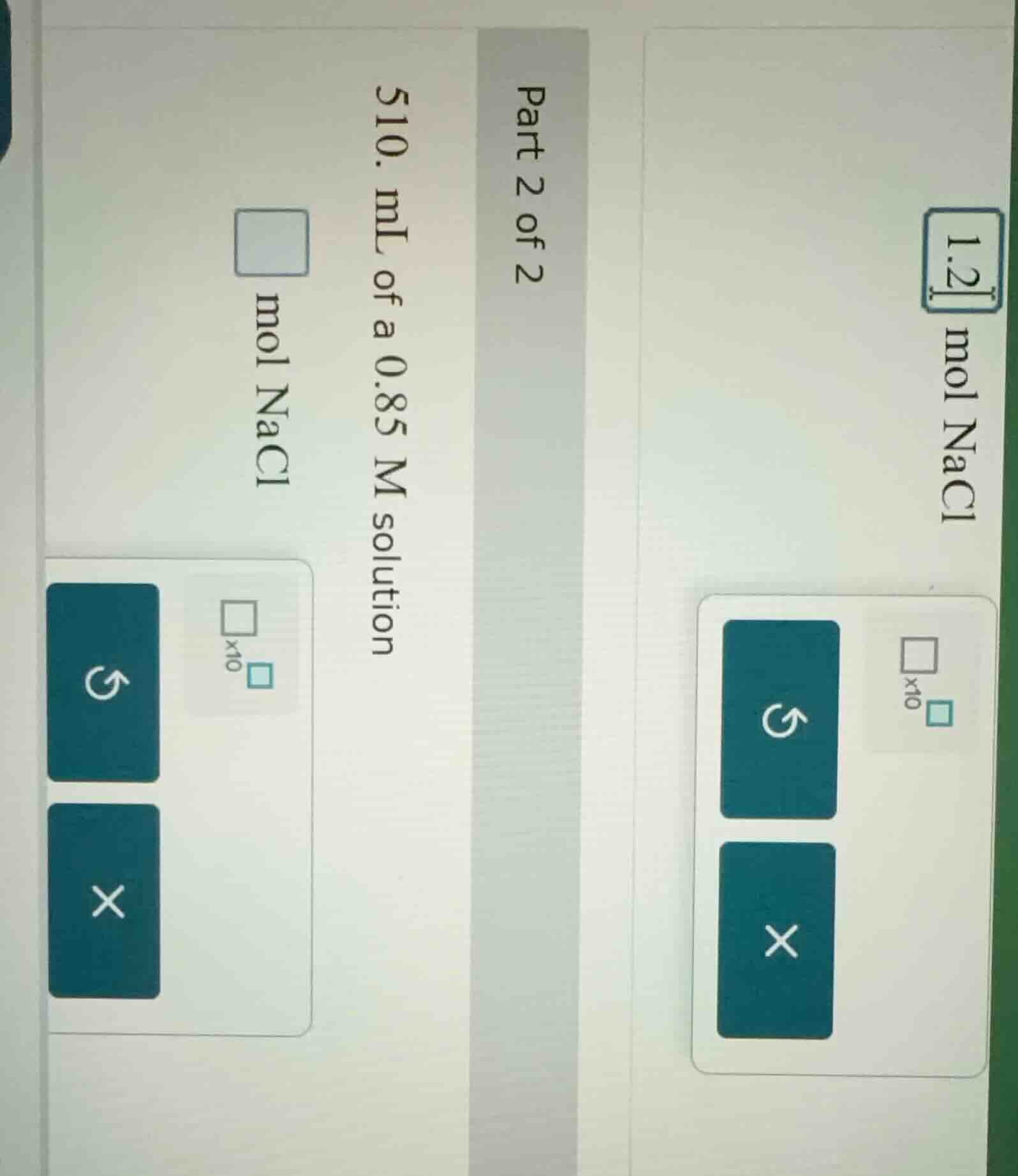
part 2 of 2
- ml of a 0.85 m solution
$square$ mol nacl
1.2 mol nacl
Step1: Convert volume to liters
$510. \text{ mL} = 510. \times \frac{1\text{ L}}{1000\text{ mL}} = 0.510\text{ L}$
Step2: Calculate moles of NaCl
Moles = Molarity × Volume (L)
$\text{Moles of NaCl} = 0.85\text{ M} \times 0.510\text{ L}$
Step3: Compute final value
$0.85 \times 0.510 = 0.4335 \approx 0.43$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.43 mol NaCl