QUESTION IMAGE
Question
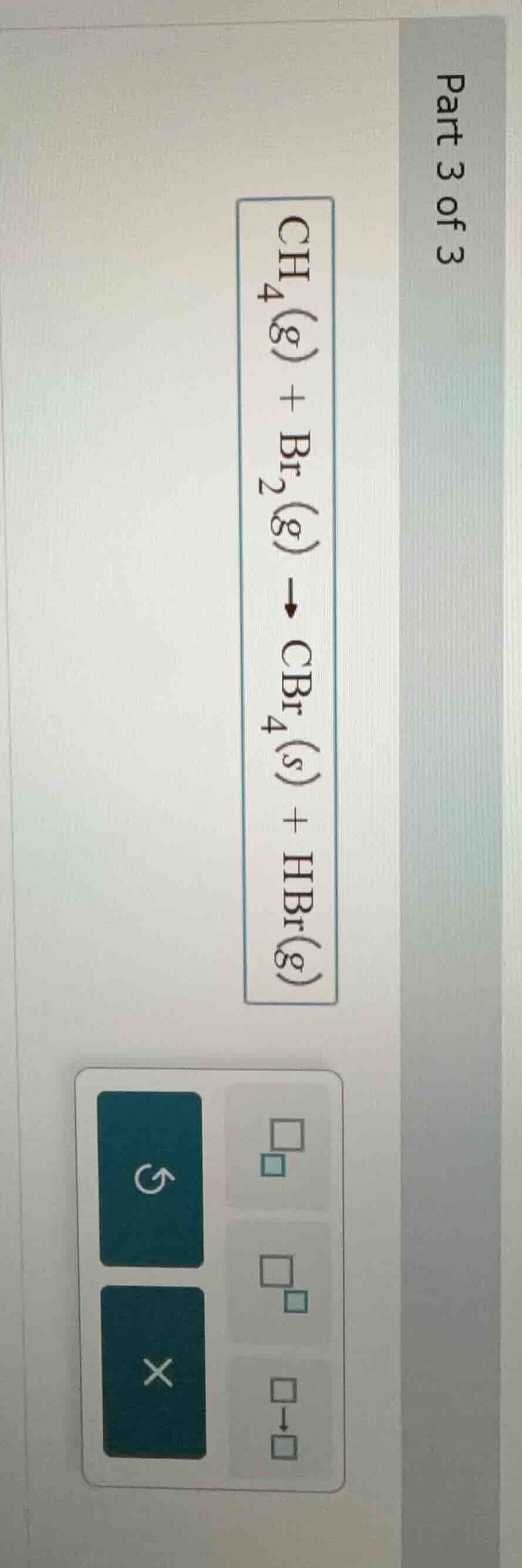
part 3 of 3 \\(\ce{ch_{4}(g) + br_{2}(g) \
ightarrow cbr_{4}(s) + hbr(g)}\\)
Step1: Balance Carbon atoms
On the left side, we have 1 C atom in \( \text{CH}_4 \). On the right side, we have 1 C atom in \( \text{CBr}_4 \). So carbon is balanced.
Step2: Balance Hydrogen atoms
On the left side, we have 4 H atoms in \( \text{CH}_4 \). On the right side, we have 1 H atom in \( \text{HBr} \). So we need to put a coefficient of 4 in front of \( \text{HBr} \) to balance H. Now the equation becomes:
\( \text{CH}_4(g) + \text{Br}_2(g)
ightarrow \text{CBr}_4(s) + 4\text{HBr}(g) \)
Step3: Balance Bromine atoms
On the right side, we have 4 Br atoms in \( \text{CBr}_4 \) and 4 Br atoms in \( 4\text{HBr} \), so total 8 Br atoms. On the left side, we have 2 Br atoms in \( \text{Br}_2 \). So we need to put a coefficient of 4 in front of \( \text{Br}_2 \) to balance Br. The balanced equation is:
\( \text{CH}_4(g) + 4\text{Br}_2(g)
ightarrow \text{CBr}_4(s) + 4\text{HBr}(g) \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The balanced chemical equation is \( \boldsymbol{\text{CH}_4(g) + 4\text{Br}_2(g)
ightarrow \text{CBr}_4(s) + 4\text{HBr}(g)} \)