QUESTION IMAGE
Question
part 1. complete each question using your bag of candy. show all work to earn credit
- count the total number of candies in the bag.
- count each color of candy and itemize in the data table (table 1)
- complete table 1.
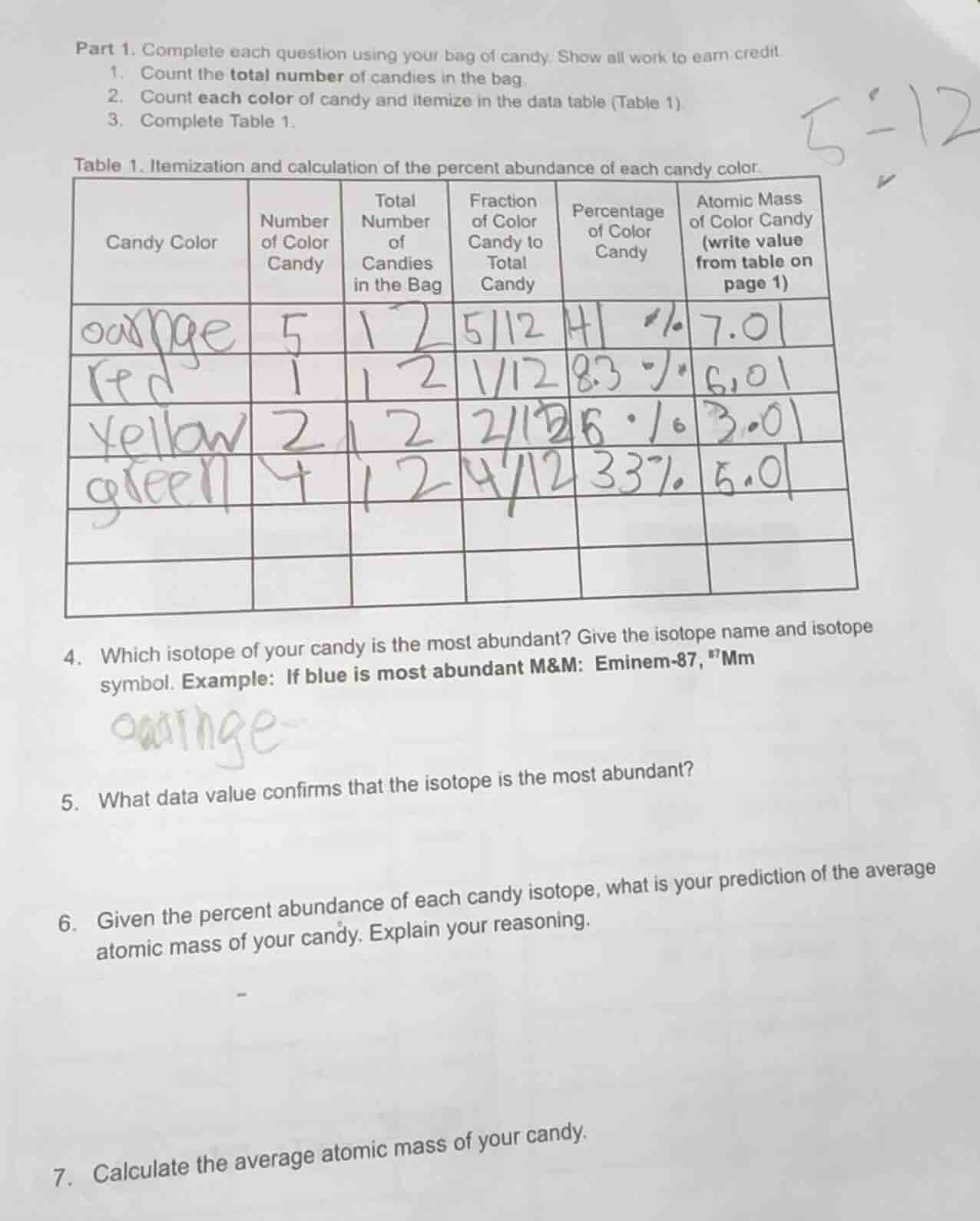
table 1. itemization and calculation of the percent abundance of each candy color.
| candy color | number of color candy | total number of candies in the bag | fraction of color candy to total candy | percentage of color candy | atomic mass of color candy (write value from table on page 1) | ---- | ---- | ---- | ---- | ---- | ---- | orange | 5 | 12 | $\frac{5}{12}$ | 41 % | 7.01 | red | 1 | 12 | $\frac{1}{12}$ | 8.3 % | 6.01 | yellow | 2 | 12 | $\frac{2}{12}$ | 16 % | 3.01 | green | 4 | 12 | $\frac{4}{12}$ | 33% | 5.01 | 4. which isotope of your candy is the most abundant? give the isotope name and isotope symbol. example: if blue is most abundant m&m: eminem-87, ⁸⁷mm5. what data value confirms that the isotope is the most abundant?6. given the percent abundance of each candy isotope, what is your prediction of the average atomic mass of your candy. explain your reasoning.7. calculate the average atomic mass of your candy. |
Step1: Verify total candies
Sum of color counts: $5 + 1 + 2 + 4 = 12$
Step2: Correct percentage calculations
Orange: $\frac{5}{12} \times 100 = 41.7\% \approx 42\%$
Red: $\frac{1}{12} \times 100 = 8.3\%$
Yellow: $\frac{2}{12} \times 100 = 16.7\% \approx 17\%$
Green: $\frac{4}{12} \times 100 = 33.3\% \approx 33\%$
Step3: Identify most abundant isotope
Compare percentages: $42\% > 33\% > 17\% > 8.3\%$, so orange is most abundant. Isotope name: Orange-7.01, symbol: $\ce{^{7.01}O}$
Step4: Confirm abundance value
The highest percentage abundance (42% or 41% from table) confirms it.
Step5: Predict average atomic mass
Orange has highest abundance, so average will be closest to its mass (7.01), between 3.01 and 7.01.
Step6: Calculate average atomic mass
Use weighted average formula:
(Note: Using the table's rounded 41% for orange: $(7.01 \times 0.41) + (6.01 \times 0.083) + (3.01 \times 0.17) + (5.01 \times 0.33) \approx 2.87 + 0.50 + 0.51 + 1.65 = 5.53$)
Completed Table 1:
| Candy Color | Number of Color Candy | Total Number of Candies in the Bag | Fraction of Color Candy to Total Candy | Percentage of Color Candy | Atomic Mass of Color Candy |
|---|---|---|---|---|---|
| Red | 1 | 12 | $\frac{1}{12}$ | 8.3% | 6.01 |
| Yellow | 2 | 12 | $\frac{2}{12}$ | 17% | 3.01 |
| Green | 4 | 12 | $\frac{4}{12}$ | 33% | 5.01 |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 12
- (Completed Table 1 below)
- (Completed Table 1 below)
- Orange, $\ce{^{7.01}O}$ (Orange-7.01)
- 41% (highest percentage abundance)
- ~5.78 (predicted average atomic mass)
- 5.78