QUESTION IMAGE
Question
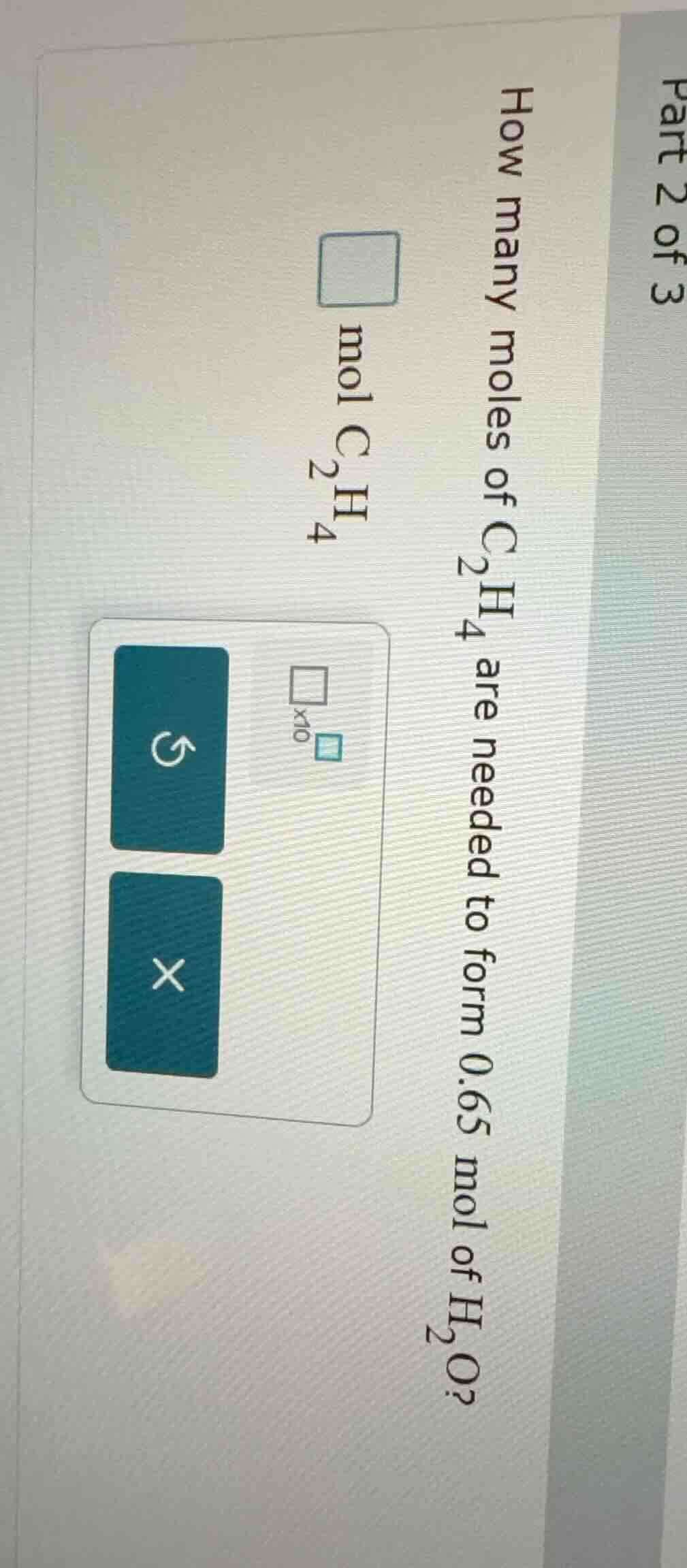
part 2 of 3 how many moles of c₂h₄ are needed to form 0.65 mol of h₂o? mol c₂h₄
To solve this, we first need the balanced chemical equation for the reaction involving \( \ce{C2H4} \) and \( \ce{H2O} \). Assuming the reaction is the hydration of ethene: \( \ce{C2H4 + H2O -> C2H5OH} \), but wait, no—wait, maybe the combustion or another reaction? Wait, no, to form \( \ce{H2O} \), maybe the combustion of \( \ce{C2H4} \). The balanced combustion equation for \( \ce{C2H4} \) is: \( \ce{C2H4 + 3O2 -> 2CO2 + 2H2O} \). Wait, no, let's check:
For \( \ce{C2H4} \) (ethene) combustion:
- Carbon: 2 on left, 2 on right (in \( \ce{CO2} \)) → so 2 \( \ce{CO2} \).
- Hydrogen: 4 on left (in \( \ce{C2H4} \)), so 2 \( \ce{H2O} \) (since 2*2=4 H).
- Oxygen: 32=6 on left (from \( \ce{O2} \)), 22 (from \( \ce{CO2} \)) + 2*1 (from \( \ce{H2O} \)) = 4 + 2 = 6. So balanced: \( \ce{C2H4 + 3O2 -> 2CO2 + 2H2O} \).
Wait, but the question is how many moles of \( \ce{C2H4} \) are needed to form 0.65 mol of \( \ce{H2O} \). From the balanced equation, the mole ratio of \( \ce{C2H4} \) to \( \ce{H2O} \) is 1:2.
Step 1: Identify the mole ratio
From \( \ce{C2H4 + 3O2 -> 2CO2 + 2H2O} \), the mole ratio of \( \ce{C2H4} \) to \( \ce{H2O} \) is \( \frac{1\ \text{mol}\ \ce{C2H4}}{2\ \text{mol}\ \ce{H2O}} \).
Step 2: Calculate moles of \( \ce{C2H4} \)
We have 0.65 mol of \( \ce{H2O} \). Using the mole ratio:
\( \text{Moles of } \ce{C2H4} = 0.65\ \text{mol}\ \ce{H2O} \times \frac{1\ \text{mol}\ \ce{C2H4}}{2\ \text{mol}\ \ce{H2O}} \)
\( \text{Moles of } \ce{C2H4} = \frac{0.65}{2} = 0.325\ \text{mol} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 0.325 \)