QUESTION IMAGE
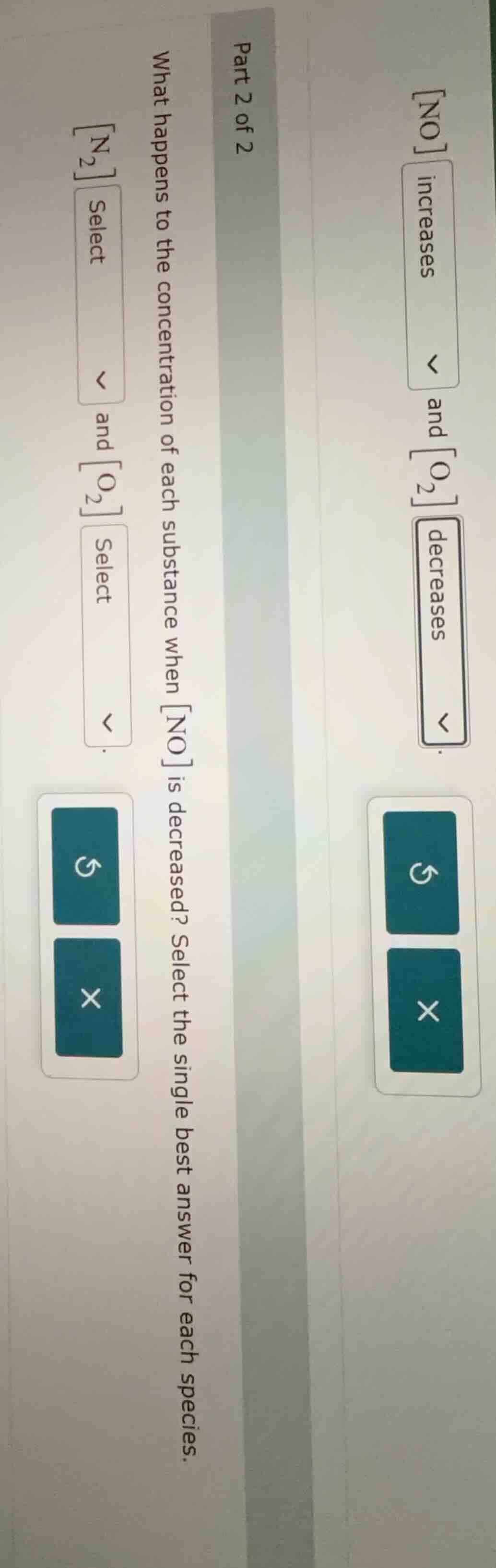
Question
part 2 of 2 what happens to the concentration of each substance when no is decreased? select the single best answer for each species. n₂ select and o₂ select
To solve this, we analyze the equilibrium reaction (likely \( \ce{N2 + O2 <=> 2NO} \)) using Le Chatelier's principle. When \([\ce{NO}]\) decreases, the system shifts to produce more \(\ce{NO}\), so it moves to the right.
For \(\boldsymbol{[\ce{N2}]}\):
Step 1: Analyze equilibrium shift
The reaction shifts right (to make \(\ce{NO}\)), so \(\ce{N2}\) (a reactant) is consumed less or produced? Wait, no—wait, the reaction is \( \ce{N2 + O2 <=> 2NO} \). If \([\ce{NO}]\) decreases, the system tries to make more \(\ce{NO}\), so it shifts right. To make more \(\ce{NO}\), \(\ce{N2}\) and \(\ce{O2}\) are consumed? Wait, no—wait, if \([\ce{NO}]\) is decreased, the equilibrium shifts to the right (toward products) to replenish \(\ce{NO}\). Wait, no: Le Chatelier’s principle says if a product (NO) is removed, the system shifts to produce more product (right), so reactants (\(\ce{N2}\), \(\ce{O2}\)) are consumed? Wait, no—wait, the reaction is \( \ce{N2 + O2 <=> 2NO} \). So if \([\ce{NO}]\) decreases, the system will shift to the right (produce more \(\ce{NO}\)), which means \(\ce{N2}\) and \(\ce{O2}\) are consumed (their concentrations decrease)? Wait, no—wait, no: if you remove a product (NO), the system shifts to make more product, so it uses up reactants (N₂, O₂) to make NO. Wait, but the question is: when \([\ce{NO}]\) is decreased, what happens to \([\ce{N2}]\) and \([\ce{O2}]\)? Wait, maybe I got the reaction wrong. Wait, maybe the reaction is \( \ce{2NO <=> N2 + O2} \)? No, that would be decomposition. Wait, the original problem’s context (from the first part) had \([\ce{NO}]\) increasing/decreasing, and \([\ce{O2}]\) decreasing. Wait, let’s re-express:
Assume the reaction is \( \ce{N2 + O2 <=> 2NO} \). If \([\ce{NO}]\) decreases, the system shifts right (to make more \(\ce{NO}\)), so \(\ce{N2}\) (reactant) is consumed (concentration decreases? No—wait, no: if you remove product (NO), the system shifts to produce more product (right), so reactants are used up? Wait, no—wait, if \([\ce{NO}]\) is low, the system wants to make more \(\ce{NO}\), so it uses \(\ce{N2}\) and \(\ce{O2}\) to make \(\ce{NO}\). Wait, no—wait, the reaction is \( \ce{N2 + O2 -> 2NO} \) (forward) and \( \ce{2NO -> N2 + O2} \) (reverse). If \([\ce{NO}]\) decreases, the reverse reaction (2NO → N₂ + O₂) slows, and the forward reaction (N₂ + O₂ → 2NO) speeds up. So to make more NO, N₂ and O₂ are consumed (their concentrations decrease)? Wait, no—wait, no: if you remove NO (product), the system shifts to the right (produce more NO), so N₂ and O₂ (reactants) are consumed, so their concentrations decrease? But that contradicts the first part. Wait, maybe the reaction is \( \ce{2NO <=> N2 + O2} \) (decomposition). Then, if \([\ce{NO}]\) decreases, the system shifts to the left (to make more NO), so N₂ and O₂ (products) are consumed, so their concentrations decrease? No, this is confusing. Wait, let’s use Le Chatelier’s principle properly:
For a reaction \( \ce{aA + bB <=> cC + dD} \), if a product (C or D) is removed, the system shifts to the right (produce more C/D), so reactants (A/B) are consumed (concentrations decrease). If a reactant (A/B) is removed, the system shifts to the left (produce more A/B), so products (C/D) are consumed (concentrations decrease).
In the first part (from the image), \([\ce{NO}]\) (product) increases or decreases? Wait, the first part’s dropdown for \([\ce{NO}]\) had “increases” and “decreases,” with “decreases” selected. Wait, no—the first part (left image) is part 2 of 2, asking about \(…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
For \(\boldsymbol{[\ce{N2}]}\): increases
For \(\boldsymbol{[\ce{O2}]}\): increases