QUESTION IMAGE
Question
pebworth, logan
sps_physical science_checkpoint2_2025-2026
23 of 40
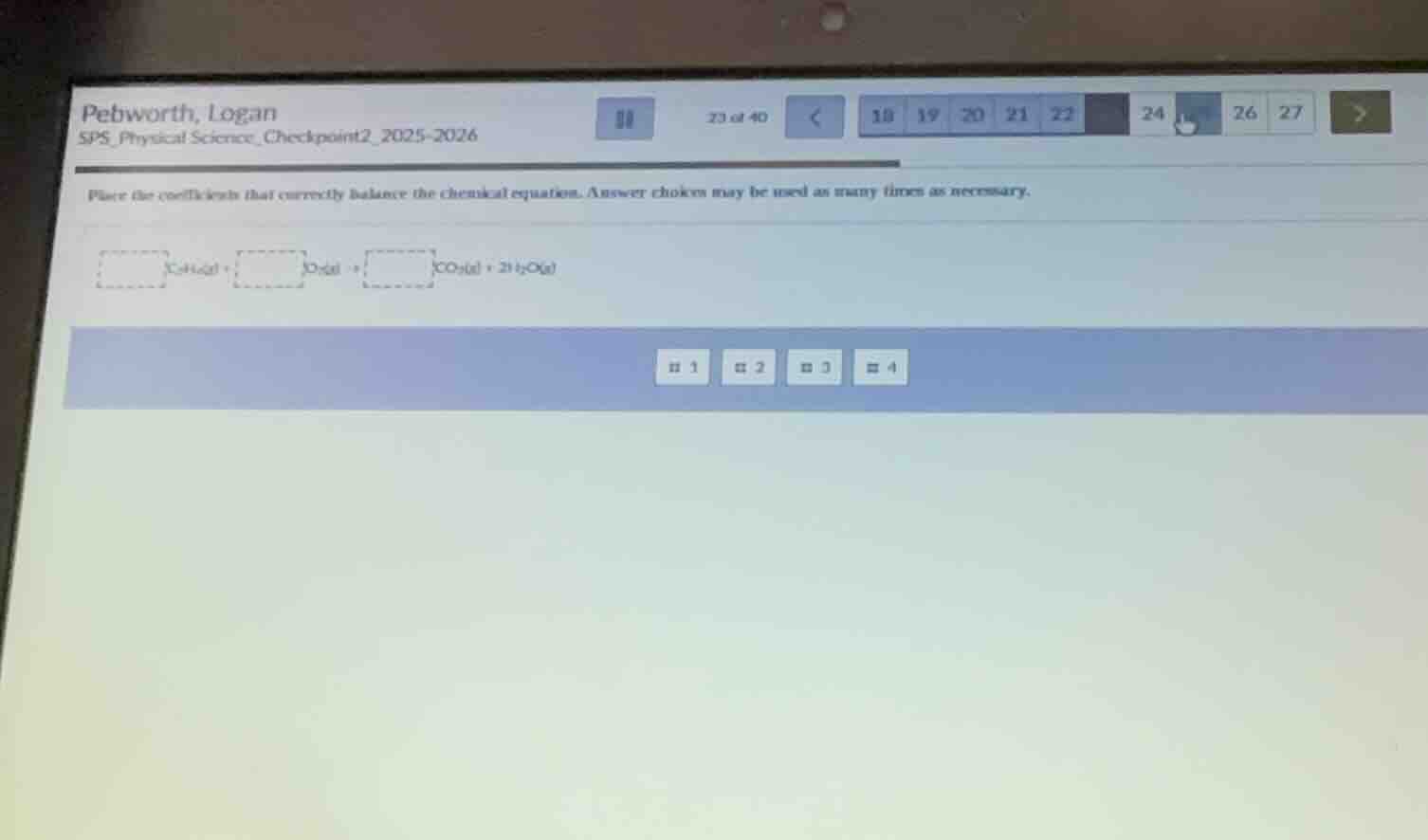
place the coefficients that correctly balance the chemical equation. answer choices may be used as many times as necessary.
c₆h₁₄(l) + o₂(g) → __ co₂(g) + 2 h₂o(g)
Step1: Balance carbon atoms
Count C on right: 2 (from $2\text{CO}_2$). Set C₂H₄ coefficient to 1: $1\text{C}_2\text{H}_4(g) + \square\text{O}_2(g)
ightarrow \square\text{CO}_2(g) + 2\text{H}_2\text{O}(g)$
Now C left = 2, so set $\text{CO}_2$ coefficient to 2: $1\text{C}_2\text{H}_4(g) + \square\text{O}_2(g)
ightarrow 2\text{CO}_2(g) + 2\text{H}_2\text{O}(g)$
Step2: Balance hydrogen atoms
Count H left: 4 (from $\text{C}_2\text{H}_4$). H right: $2\times2=4$ (from $2\text{H}_2\text{O}$), so H is balanced.
Step3: Balance oxygen atoms
Count O right: $(2\times2)+(2\times1)=6$. Divide by 2 (per $\text{O}_2$ molecule) to get $\text{O}_2$ coefficient: $\frac{6}{2}=3$. Final equation: $1\text{C}_2\text{H}_4(g) + 3\text{O}_2(g)
ightarrow 2\text{CO}_2(g) + 2\text{H}_2\text{O}(g)$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1, 3, 2 (in order for $\text{C}_2\text{H}_4$, $\text{O}_2$, $\text{CO}_2$)