QUESTION IMAGE
Question
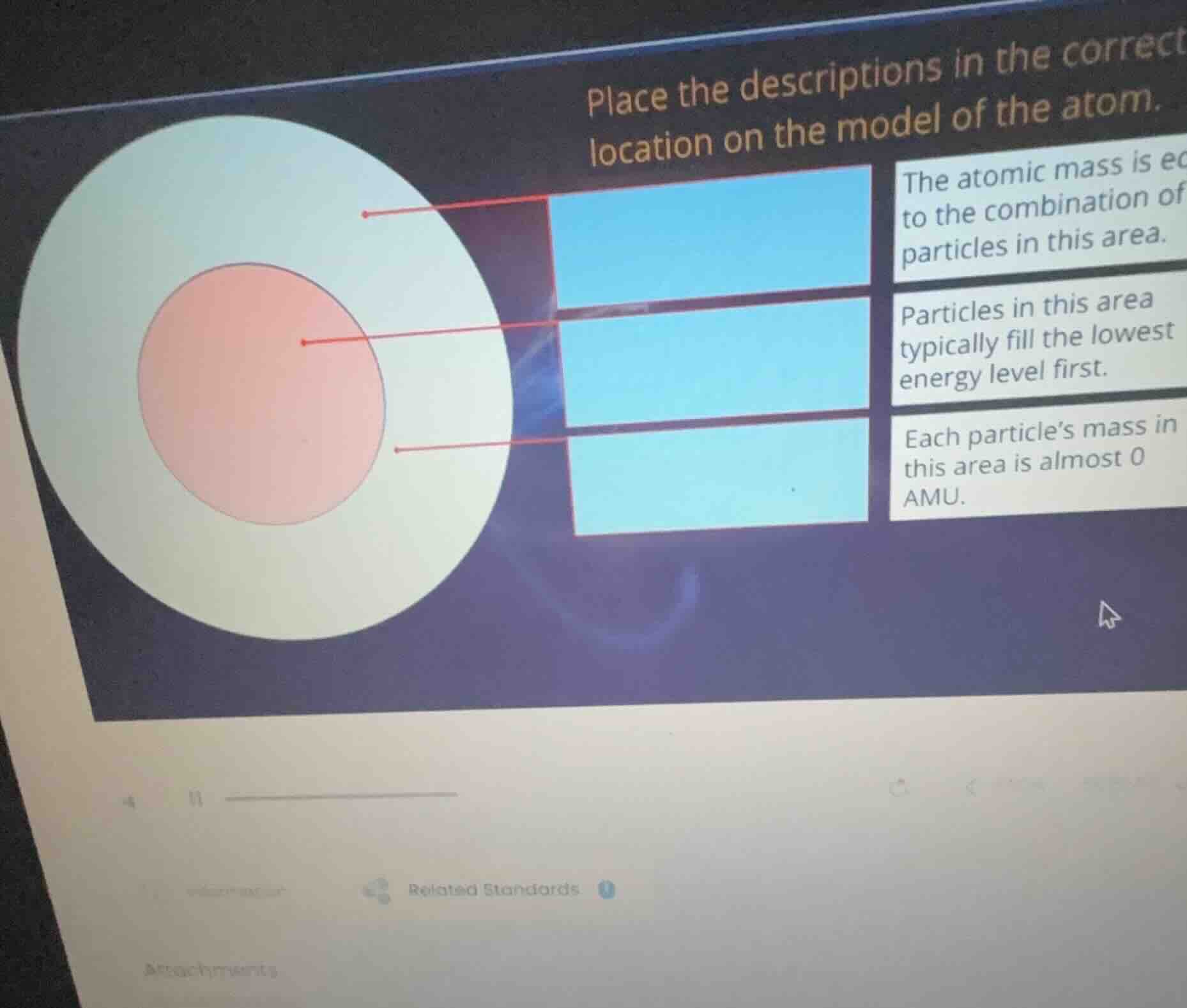
place the descriptions in the correct location on the model of the atom. the atomic mass is eq to the combination of particles in this area. particles in this area typically fill the lowest energy level first. each particle’s mass in this area is almost 0 amu.
Brief Explanations
- For the outermost (electron cloud) region: Particles here are electrons. Electrons have a mass of ~0 AMU, so "Each particle’s mass in this area is almost 0 AMU" and "Particles in this area typically fill the lowest energy level first" (electrons fill energy levels from lowest to highest) apply. But let's match each description:
- The atomic mass is due to protons + neutrons (in the nucleus), so "The atomic mass is equal to the combination of particles in this area" goes to the nucleus (the pink circle).
- "Particles in this area typically fill the lowest energy level first" refers to electrons (in the electron cloud, the light - colored outer region) as electrons fill energy levels starting from the lowest.
- "Each particle’s mass in this area is almost 0 AMU" also refers to electrons (in the electron cloud) since electrons have negligible mass.
- Let's assign each description to the correct part of the atom model:
- The pink circle is the nucleus (contains protons and neutrons, which contribute to atomic mass). So "The atomic mass is equal to the combination of particles in this area" is for the nucleus (the middle red dot - pointing area).
- The light - colored outer region is the electron cloud. "Particles in this area typically fill the lowest energy level first" (electrons filling energy levels) and "Each particle’s mass in this area is almost 0 AMU" (electrons' mass) are for the electron cloud. If we assume the three red - pointed areas are (from top to bottom): electron cloud, nucleus, electron cloud (or maybe a middle layer? Wait, the standard atom model has nucleus (center) and electron cloud (outer). Let's re - check:
- Nucleus (center, pink): contains protons and neutrons, so atomic mass (protons + neutrons) is from here. So "The atomic mass is equal to the combination of particles in this area" → nucleus (middle red dot).
- Electron cloud (outer, light - colored): electrons are here. Electrons fill lowest energy levels first (Aufbau principle) and have ~0 AMU mass. So "Particles in this area typically fill the lowest energy level first" → electron cloud (top red dot area). "Each particle’s mass in this area is almost 0 AMU" → electron cloud (bottom red dot area).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Top (outer region, electron cloud): "Particles in this area typically fill the lowest energy level first" and "Each particle’s mass in this area is almost 0 AMU" (either can be placed here, but based on energy levels first for filling, "Particles in this area typically fill the lowest energy level first" is a better fit for the electron cloud's electron behavior).
- Middle (nucleus): "The atomic mass is equal to the combination of particles in this area".
- Bottom (outer region, electron cloud): "Each particle’s mass in this area is almost 0 AMU" (if top is for energy level filling).
(If we consider the three regions as: outer electron cloud (top), nucleus (middle), and maybe a middle - outer or another part, but the standard model has nucleus and electron cloud. So the correct assignments are:
- Middle (nucleus): "The atomic mass is equal to the combination of particles in this area".
- Top (electron cloud): "Particles in this area typically fill the lowest energy level first".
- Bottom (electron cloud): "Each particle’s mass in this area is almost 0 AMU".)