QUESTION IMAGE
Question
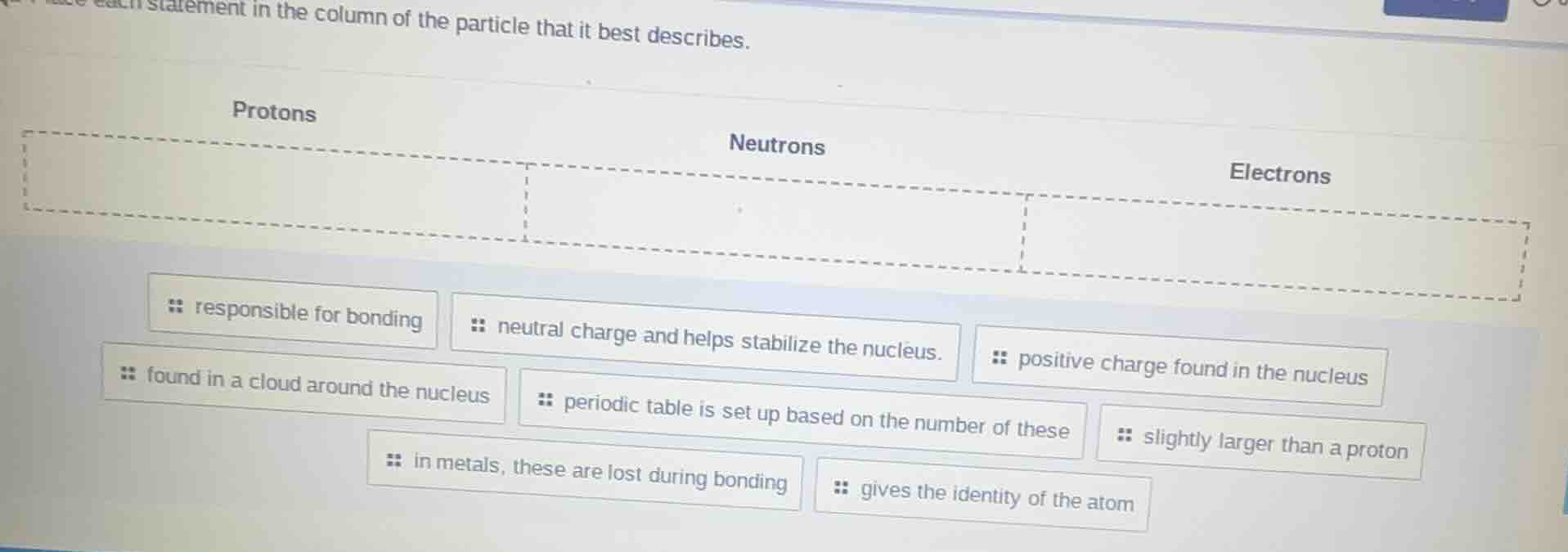
place each statement in the column of the particle that it best describes.
protons
neutrons
electrons
- responsible for bonding
- neutral charge and helps stabilize the nucleus.
- positive charge found in the nucleus
- found in a cloud around the nucleus
- periodic table is set up based on the number of these
- slightly larger than a proton
- in metals, these are lost during bonding
- gives the identity of the atom
Brief Explanations
- Protons: Define an atom's identity, have a positive nuclear charge, and the periodic table is ordered by their count.
- Neutrons: Are neutral, stabilize the atomic nucleus, and have a slightly larger mass than protons.
- Electrons: Orbit the nucleus in a cloud, drive chemical bonding, and are lost by metals during bonding.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Protons
- positive charge found in the nucleus
- periodic table is set up based on the number of these
- gives the identity of the atom
Neutrons
- neutral charge and helps stabilize the nucleus
- slightly larger than a proton
Electrons
- responsible for bonding
- found in a cloud around the nucleus
- in metals, these are lost during bonding