QUESTION IMAGE
Question
practice: isotope calculations #2
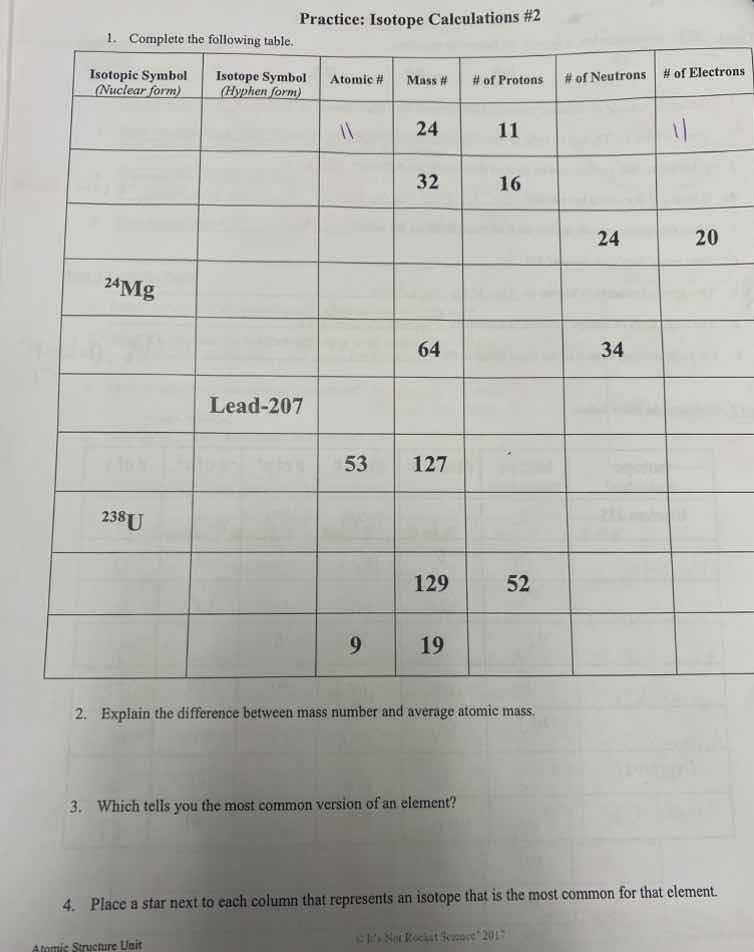
- complete the following table.
| isotopic symbol (nuclear form) | isotope symbol (hyphen form) | atomic # | mass # | # of protons | # of neutrons | # of electrons |
|---|---|---|---|---|---|---|
| 32 | 16 | |||||
| 24 | 20 | |||||
| $^{24}$mg | ||||||
| 64 | 34 | |||||
| lead-207 | ||||||
| 53 | 127 | |||||
| $^{238}$u | ||||||
| 129 | 52 | |||||
| 9 | 19 |
- explain the difference between mass number and average atomic mass.
- which tells you the most common version of an element?
- place a star next to each column that represents an isotope that is the most common for that element.
atomic structure unit © 2017 star rocket science
Part 1: Completing the Table
To complete the table, we use the following relationships:
- Atomic Number (\(Z\)) = Number of Protons = Number of Electrons (in neutral atoms).
- Mass Number (\(A\)) = Number of Protons + Number of Neutrons.
- Isotopic Symbol (Nuclear Form): \(\ce{^A_ZX}\) (where \(X\) is the element symbol).
- Isotopic Symbol (Hyphen Form): \(X\text{-}A\).
Row 1:
- Atomic # = 11 (Na, Sodium), Mass # = 24, Protons = 11, Electrons = 11.
- Neutrons = \(24 - 11 = 13\).
- Nuclear Form: \(\ce{^{24}_{11}Na}\), Hyphen Form: Sodium-24.
Row 2:
- Protons = 16 (S, Sulfur), Mass # = 32, Atomic # = 16.
- Neutrons = \(32 - 16 = 16\), Electrons = 16.
- Nuclear Form: \(\ce{^{32}_{16}S}\), Hyphen Form: Sulfur-32.
Row 3:
- Neutrons = 24, Electrons = 20 (so Protons = 20, Atomic # = 20, Ca, Calcium).
- Mass # = \(20 + 24 = 44\).
- Nuclear Form: \(\ce{^{44}_{20}Ca}\), Hyphen Form: Calcium-44.
Row 4: \(\ce{^{24}Mg}\)
- Mg (Magnesium) has Atomic # = 12 (Protons = 12, Electrons = 12).
- Mass # = 24, Neutrons = \(24 - 12 = 12\).
- Hyphen Form: Magnesium-24.
Row 5:
- Mass # = 64, Neutrons = 34, so Protons = \(64 - 34 = 30\) (Zn, Zinc, Atomic # = 30, Electrons = 30).
- Nuclear Form: \(\ce{^{64}_{30}Zn}\), Hyphen Form: Zinc-64.
Row 6: Lead-207
- Lead (Pb) has Atomic # = 82 (Protons = 82, Electrons = 82).
- Mass # = 207, Neutrons = \(207 - 82 = 125\).
- Nuclear Form: \(\ce{^{207}_{82}Pb}\).
Row 7:
- Atomic # = 53 (I, Iodine), Mass # = 127, Protons = 53, Electrons = 53.
- Neutrons = \(127 - 53 = 74\).
- Nuclear Form: \(\ce{^{127}_{53}I}\), Hyphen Form: Iodine-127.
Row 8: \(\ce{^{238}U}\)
- U (Uranium) has Atomic # = 92 (Protons = 92, Electrons = 92).
- Mass # = 238, Neutrons = \(238 - 92 = 146\).
- Hyphen Form: Uranium-238.
Row 9:
- Mass # = 129, Protons = 52 (Te, Tellurium, Atomic # = 52, Electrons = 52).
- Neutrons = \(129 - 52 = 77\).
- Nuclear Form: \(\ce{^{129}_{52}Te}\), Hyphen Form: Tellurium-129.
Row 10:
- Atomic # = 9 (F, Fluorine), Mass # = 19, Protons = 9, Electrons = 9.
- Neutrons = \(19 - 9 = 10\).
- Nuclear Form: \(\ce{^{19}_{9}F}\), Hyphen Form: Fluorine-19.
Part 2: Mass Number vs. Average Atomic Mass
- Mass Number: Integer (protons + neutrons) for a single isotope (e.g., Carbon-12 has mass number 12).
- Average Atomic Mass: Weighted average of masses of all naturally occurring isotopes of an element, in amu (e.g., average atomic mass of carbon is ~12.011 amu).
Part 3: Most Common Isotope Indicator
The average atomic mass (from the periodic table) tells you the most common isotope (since it is weighted by abundance). For example, carbon’s average atomic mass (~12.01) is closest to Carbon-12, so Carbon-12 is the most common.
Part 4: Identifying Most Common Isotopes
A star (\(*\)) is placed next to isotopes where the mass number is closest to the element’s average atomic mass (from the periodic table):
- \(\ce{^{24}Mg}\) (Mg: avg ~24.305) → *
- \(\ce{^{127}_{53}I}\) (I: avg ~126.90) → *
- \(\ce{^{19}_{9}F}\) (F: avg ~18.998) → *
- \(\ce{^{32}_{16}S}\) (S: avg ~32.06) → *
Final Table (Key Cells Filled):
| Isotopic Symbol (Nuclear) | Isotope Symbol (Hyphen) | Atomic # | Mass # | # Protons | # Neutrons | # Electrons |
|---|---|---|---|---|---|---|
| \(\ce{^{32}_{16}S}\) | Sulfur-32 | 16 | 32 | 16 | … |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part 1: Completing the Table
To complete the table, we use the following relationships:
- Atomic Number (\(Z\)) = Number of Protons = Number of Electrons (in neutral atoms).
- Mass Number (\(A\)) = Number of Protons + Number of Neutrons.
- Isotopic Symbol (Nuclear Form): \(\ce{^A_ZX}\) (where \(X\) is the element symbol).
- Isotopic Symbol (Hyphen Form): \(X\text{-}A\).
Row 1:
- Atomic # = 11 (Na, Sodium), Mass # = 24, Protons = 11, Electrons = 11.
- Neutrons = \(24 - 11 = 13\).
- Nuclear Form: \(\ce{^{24}_{11}Na}\), Hyphen Form: Sodium-24.
Row 2:
- Protons = 16 (S, Sulfur), Mass # = 32, Atomic # = 16.
- Neutrons = \(32 - 16 = 16\), Electrons = 16.
- Nuclear Form: \(\ce{^{32}_{16}S}\), Hyphen Form: Sulfur-32.
Row 3:
- Neutrons = 24, Electrons = 20 (so Protons = 20, Atomic # = 20, Ca, Calcium).
- Mass # = \(20 + 24 = 44\).
- Nuclear Form: \(\ce{^{44}_{20}Ca}\), Hyphen Form: Calcium-44.
Row 4: \(\ce{^{24}Mg}\)
- Mg (Magnesium) has Atomic # = 12 (Protons = 12, Electrons = 12).
- Mass # = 24, Neutrons = \(24 - 12 = 12\).
- Hyphen Form: Magnesium-24.
Row 5:
- Mass # = 64, Neutrons = 34, so Protons = \(64 - 34 = 30\) (Zn, Zinc, Atomic # = 30, Electrons = 30).
- Nuclear Form: \(\ce{^{64}_{30}Zn}\), Hyphen Form: Zinc-64.
Row 6: Lead-207
- Lead (Pb) has Atomic # = 82 (Protons = 82, Electrons = 82).
- Mass # = 207, Neutrons = \(207 - 82 = 125\).
- Nuclear Form: \(\ce{^{207}_{82}Pb}\).
Row 7:
- Atomic # = 53 (I, Iodine), Mass # = 127, Protons = 53, Electrons = 53.
- Neutrons = \(127 - 53 = 74\).
- Nuclear Form: \(\ce{^{127}_{53}I}\), Hyphen Form: Iodine-127.
Row 8: \(\ce{^{238}U}\)
- U (Uranium) has Atomic # = 92 (Protons = 92, Electrons = 92).
- Mass # = 238, Neutrons = \(238 - 92 = 146\).
- Hyphen Form: Uranium-238.
Row 9:
- Mass # = 129, Protons = 52 (Te, Tellurium, Atomic # = 52, Electrons = 52).
- Neutrons = \(129 - 52 = 77\).
- Nuclear Form: \(\ce{^{129}_{52}Te}\), Hyphen Form: Tellurium-129.
Row 10:
- Atomic # = 9 (F, Fluorine), Mass # = 19, Protons = 9, Electrons = 9.
- Neutrons = \(19 - 9 = 10\).
- Nuclear Form: \(\ce{^{19}_{9}F}\), Hyphen Form: Fluorine-19.
Part 2: Mass Number vs. Average Atomic Mass
- Mass Number: Integer (protons + neutrons) for a single isotope (e.g., Carbon-12 has mass number 12).
- Average Atomic Mass: Weighted average of masses of all naturally occurring isotopes of an element, in amu (e.g., average atomic mass of carbon is ~12.011 amu).
Part 3: Most Common Isotope Indicator
The average atomic mass (from the periodic table) tells you the most common isotope (since it is weighted by abundance). For example, carbon’s average atomic mass (~12.01) is closest to Carbon-12, so Carbon-12 is the most common.
Part 4: Identifying Most Common Isotopes
A star (\(*\)) is placed next to isotopes where the mass number is closest to the element’s average atomic mass (from the periodic table):
- \(\ce{^{24}Mg}\) (Mg: avg ~24.305) → *
- \(\ce{^{127}_{53}I}\) (I: avg ~126.90) → *
- \(\ce{^{19}_{9}F}\) (F: avg ~18.998) → *
- \(\ce{^{32}_{16}S}\) (S: avg ~32.06) → *
Final Table (Key Cells Filled):
| Isotopic Symbol (Nuclear) | Isotope Symbol (Hyphen) | Atomic # | Mass # | # Protons | # Neutrons | # Electrons | |
|---|---|---|---|---|---|---|---|
| \(\ce{^{32}_{16}S}\) | Sulfur-32 | 16 | 32 | 16 | 16 | 16 | |
| \(\ce{^{44}_{20}Ca}\) | Calcium-44 | 20 | 44 | 20 | 24 | 20 | |
| \(\ce{^{24}Mg}\) | Magnesium-24 | 12 | 24 | 12 | 12 | 12 | * |
| \(\ce{^{64}_{30}Zn}\) | Zinc-64 | 30 | 64 | 30 | 34 | 30 | |
| \(\ce{^{207}_{82}Pb}\) | Lead-207 | 82 | 207 | 82 | 125 | 82 | |
| \(\ce{^{127}_{53}I}\) | Iodine-127 | 53 | 127 | 53 | 74 | 53 | * |
| \(\ce{^{238}U}\) | Uranium-238 | 92 | 238 | 92 | 146 | 92 | |
| \(\ce{^{129}_{52}Te}\) | Tellurium-129 | 52 | 129 | 52 | 77 | 52 | |
| \(\ce{^{19}_{9}F}\) | Fluorine-19 | 9 | 19 | 9 | 10 | 9 | * |
(Note: Stars are placed based on average atomic mass proximity.)