QUESTION IMAGE
Question
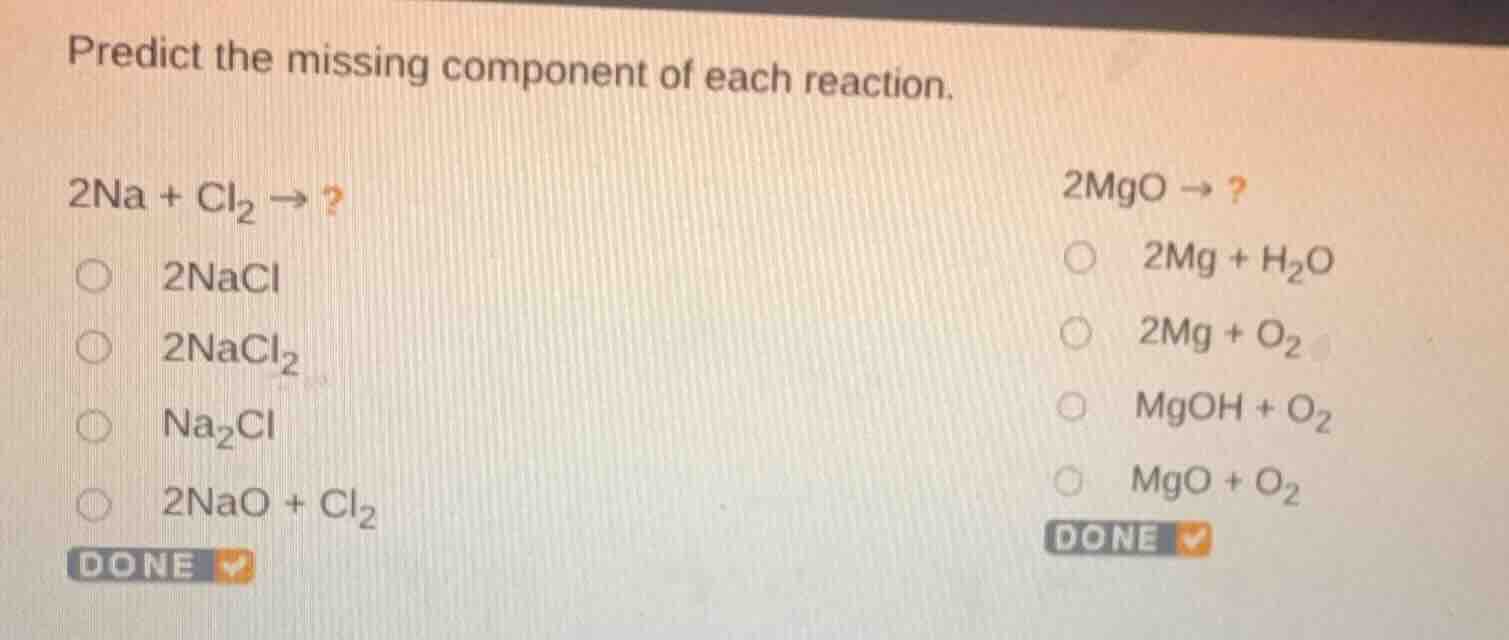
predict the missing component of each reaction.\
$2\ce{na} + \ce{cl_{2}} \
ightarrow$ ?\
$\circ$ $2\ce{nacl}$\
$\circ$ $2\ce{nacl_{2}}$\
$\circ$ $\ce{na_{2}cl}$\
$\circ$ $2\ce{nao} + \ce{cl_{2}}$\
done\
$2\ce{mgo} \
ightarrow$ ?\
$\circ$ $2\ce{mg} + \ce{h_{2}o}$\
$\circ$ $2\ce{mg} + \ce{o_{2}}$\
$\circ$ $\ce{mgoh} + \ce{o_{2}}$\
$\circ$ $\ce{mgo} + \ce{o_{2}}$\
done
First Reaction ($\boldsymbol{2\text{Na} + \text{Cl}_2
ightarrow ?}$)
Step1: Identify Reaction Type
This is a synthesis reaction (combination reaction) where sodium (Na) and chlorine gas ($\text{Cl}_2$) combine. Sodium has a charge of $+1$, chlorine has a charge of $-1$.
Step2: Apply Chemical Bonding
To form a neutral compound, one Na (${+1}$) combines with one Cl (${-1}$), so the formula is $\text{NaCl}$. From the reaction $2\text{Na} + \text{Cl}_2$, the stoichiometry gives $2\text{NaCl}$ (since 2 Na atoms react with 1 $\text{Cl}_2$ molecule, producing 2 $\text{NaCl}$ units).
Second Reaction ($\boldsymbol{2\text{MgO}
ightarrow ?}$)
Step1: Identify Reaction Type
This is a decomposition reaction (compound breaks down). $\text{MgO}$ (magnesium oxide) decomposes. Magnesium oxide is made of Mg and O.
Step2: Predict Products
Decomposing $\text{MgO}$ should give its constituent elements. The formula for magnesium is $\text{Mg}$ (elemental state, solid) and oxygen is $\text{O}_2$ (gaseous diatomic form). From $2\text{MgO}$, the decomposition would be $2\text{Mg} + \text{O}_2$ (balancing: 2 Mg from 2 $\text{MgO}$, and 2 O from 2 $\text{MgO}$ form 1 $\text{O}_2$ molecule).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
For $2\text{Na} + \text{Cl}_2
ightarrow ?$: $\boldsymbol{2\text{NaCl}}$
For $2\text{MgO}
ightarrow ?$: $\boldsymbol{2\text{Mg} + \text{O}_2}$