QUESTION IMAGE
Question
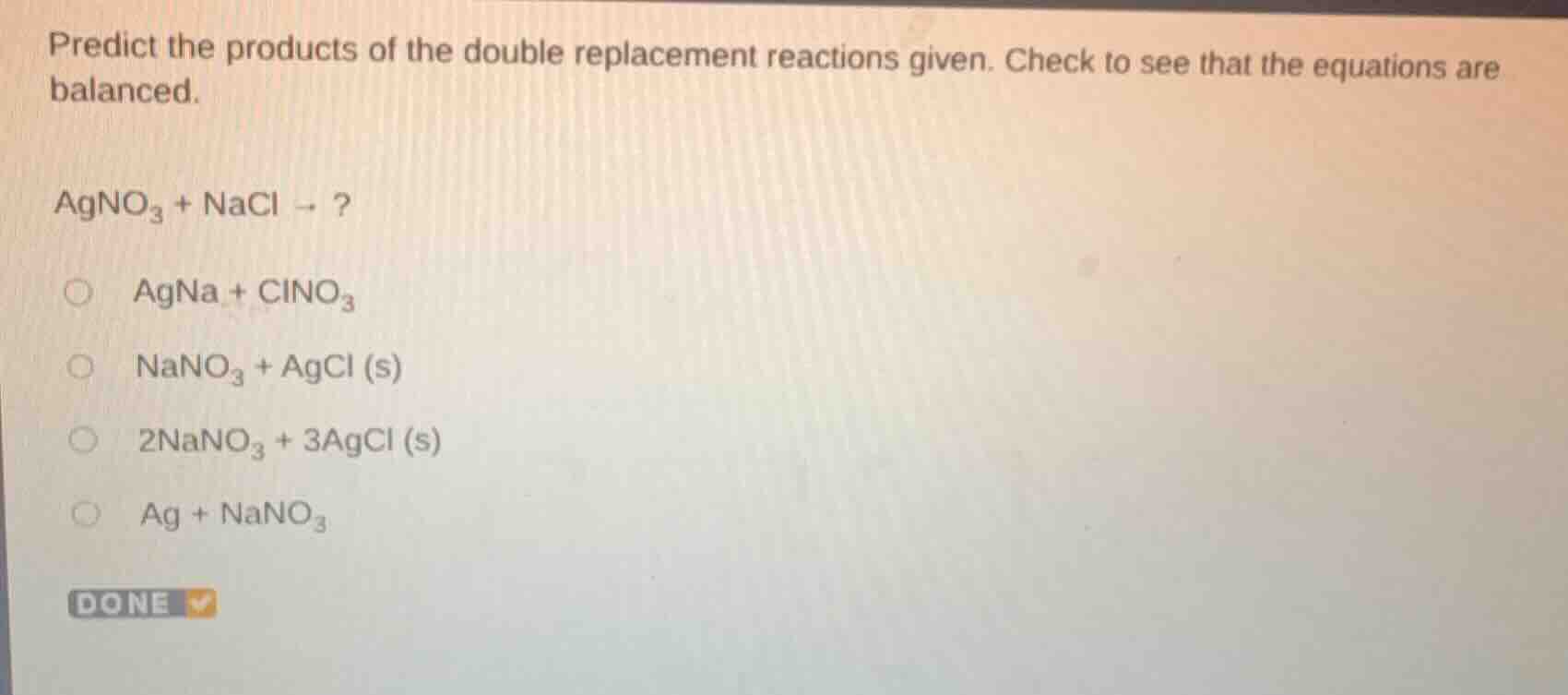
predict the products of the double replacement reactions given. check to see that the equations are balanced.
agno₃ + nacl → ?
agna + clno₃
nano₃ + agcl (s)
2nano₃ + 3agcl (s)
ag + nano₃
In a double - replacement reaction, the cations and anions of the two reactants switch places. For the reaction between $\ce{AgNO_3}$ and $\ce{NaCl}$, the silver ion ($\ce{Ag+}$) from $\ce{AgNO_3}$ combines with the chloride ion ($\ce{Cl-}$) from $\ce{NaCl}$ to form silver chloride ($\ce{AgCl}$), which is a solid (precipitate). The sodium ion ($\ce{Na+}$) from $\ce{NaCl}$ combines with the nitrate ion ($\ce{NO_3-}$) from $\ce{AgNO_3}$ to form sodium nitrate ($\ce{NaNO_3}$). The balanced equation is $\ce{AgNO_3 + NaCl = NaNO_3 + AgCl(s)}$. The first option has incorrect compound formation. The third option has incorrect stoichiometry. The fourth option is a single - replacement - like reaction, not double - replacement.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\ce{NaNO_3 + AgCl (s)}$