QUESTION IMAGE
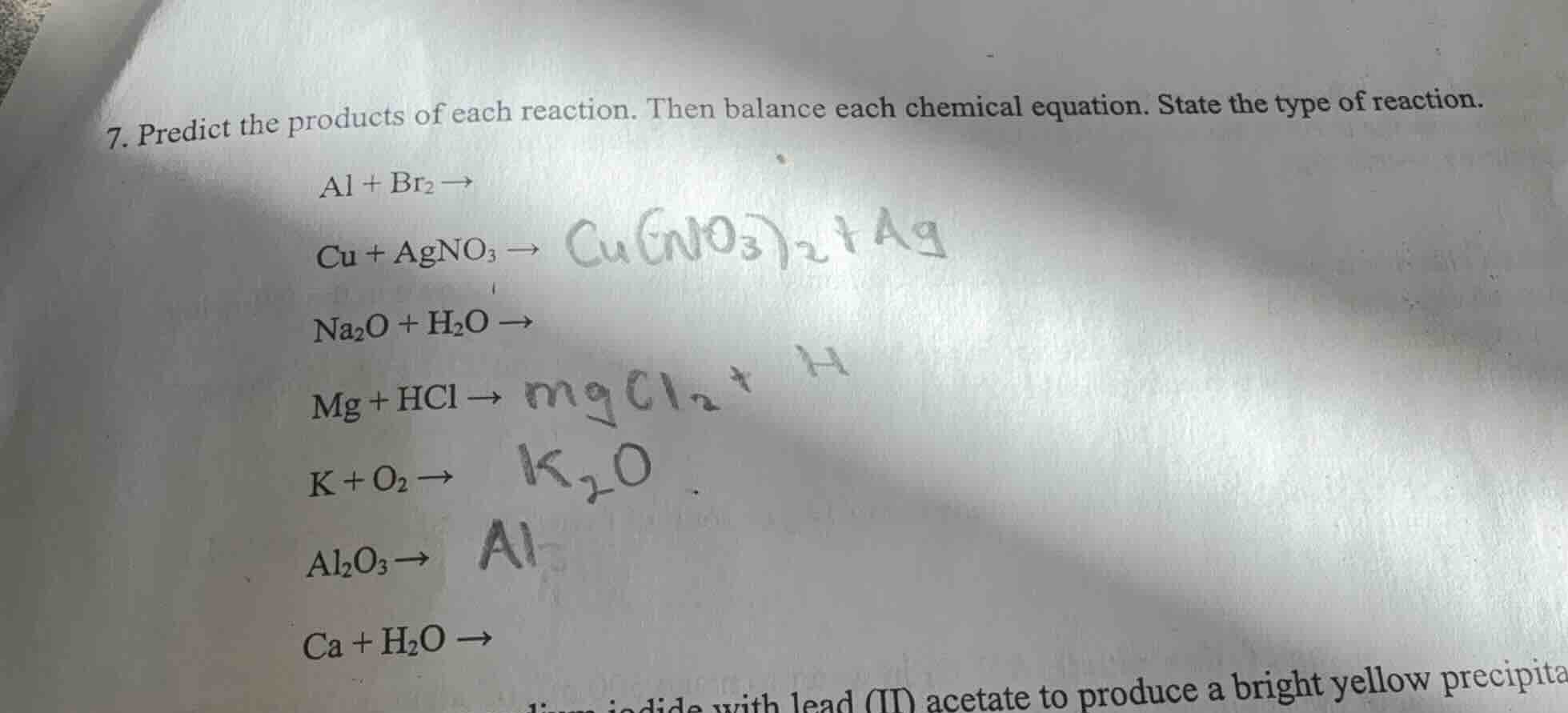
Question
- predict the products of each reaction. then balance each chemical equation. state the type of reaction.
$ce{al + br_{2} ->}$
$ce{cu + agno_{3} ->}$
$ce{na_{2}o + h_{2}o ->}$
$ce{mg + hcl ->}$
$ce{k + o_{2} ->}$
$ce{al_{2}o_{3} ->}$
$ce{ca + h_{2}o ->}$
with lead (ii) acetate to produce a bright yellow precipita
1. Reaction: $\boldsymbol{\ce{Al + Br_{2}
ightarrow}}$
Step 1: Predict Products (Synthesis Reaction)
Aluminum (Al) reacts with bromine ($\ce{Br_{2}}$) to form aluminum bromide ($\ce{AlBr_{3}}$) (synthesis: metal + non - metal $
ightarrow$ ionic compound).
Unbalanced: $\ce{Al + Br_{2}
ightarrow AlBr_{3}}$
Step 2: Balance the Equation
- Balance Br: There are 2 Br on left, 3 on right. LCM of 2 and 3 is 6. So put 3 in front of $\ce{Br_{2}}$ and 2 in front of $\ce{AlBr_{3}}$.
$\ce{Al + 3Br_{2}
ightarrow 2AlBr_{3}}$
- Balance Al: Now there are 2 Al on right, so put 2 in front of Al.
$\ce{2Al + 3Br_{2}
ightarrow 2AlBr_{3}}$
Step 3: Reaction Type
Synthesis (combination) reaction (two substances combine to form one).
2. Reaction: $\boldsymbol{\ce{Cu + AgNO_{3}
ightarrow}}$
Step 1: Predict Products (Single - Displacement)
Copper (Cu) is more reactive than silver (Ag), so Cu displaces Ag from $\ce{AgNO_{3}}$. Products are $\ce{Cu(NO_{3})_{2}}$ and Ag.
Unbalanced: $\ce{Cu + AgNO_{3}
ightarrow Cu(NO_{3})_{2} + Ag}$
Step 2: Balance the Equation
- Balance $\ce{NO_{3}^{-}}$: 2 on right, so put 2 in front of $\ce{AgNO_{3}}$.
$\ce{Cu + 2AgNO_{3}
ightarrow Cu(NO_{3})_{2} + Ag}$
- Balance Ag: 2 on left, so put 2 in front of Ag.
$\ce{Cu + 2AgNO_{3}
ightarrow Cu(NO_{3})_{2} + 2Ag}$
Step 3: Reaction Type
Single - displacement reaction (one element displaces another in a compound).
3. Reaction: $\boldsymbol{\ce{Na_{2}O + H_{2}O
ightarrow}}$
Step 1: Predict Products (Synthesis)
Sodium oxide ($\ce{Na_{2}O}$) reacts with water ($\ce{H_{2}O}$) to form sodium hydroxide ($\ce{NaOH}$) (synthesis of a base from metal oxide and water).
Unbalanced: $\ce{Na_{2}O + H_{2}O
ightarrow NaOH}$
Step 2: Balance the Equation
- Balance Na: 2 on left, so put 2 in front of $\ce{NaOH}$.
$\ce{Na_{2}O + H_{2}O
ightarrow 2NaOH}$
- Check O and H: O: 2 (left: 1 in $\ce{Na_{2}O}$ + 1 in $\ce{H_{2}O}$ = 2; right: 2 in 2$\ce{NaOH}$). H: 2 (left: 2 in $\ce{H_{2}O}$; right: 2 in 2$\ce{NaOH}$). Balanced.
Step 3: Reaction Type
Synthesis (combination) reaction (metal oxide + water $
ightarrow$ base).
4. Reaction: $\boldsymbol{\ce{Mg + HCl
ightarrow}}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s (with balanced equations and reaction types):
- $\ce{2Al + 3Br_{2}
ightarrow 2AlBr_{3}}$; Type: Synthesis
- $\ce{Cu + 2AgNO_{3}
ightarrow Cu(NO_{3})_{2} + 2Ag}$; Type: Single - Displacement
- $\ce{Na_{2}O + H_{2}O
ightarrow 2NaOH}$; Type: Synthesis
- $\ce{Mg + 2HCl
ightarrow MgCl_{2} + H_{2}\uparrow}$; Type: Single - Displacement
- $\ce{2K + O_{2}
ightarrow K_{2}O_{2}}$ (or $\ce{4K + O_{2}
ightarrow 2K_{2}O}$); Type: Synthesis
- $\ce{2Al_{2}O_{3}
ightarrow 4Al + 3O_{2}\uparrow}$; Type: Decomposition
- $\ce{Ca + 2H_{2}O
ightarrow Ca(OH)_{2} + H_{2}\uparrow}$; Type: Single - Displacement