QUESTION IMAGE
Question
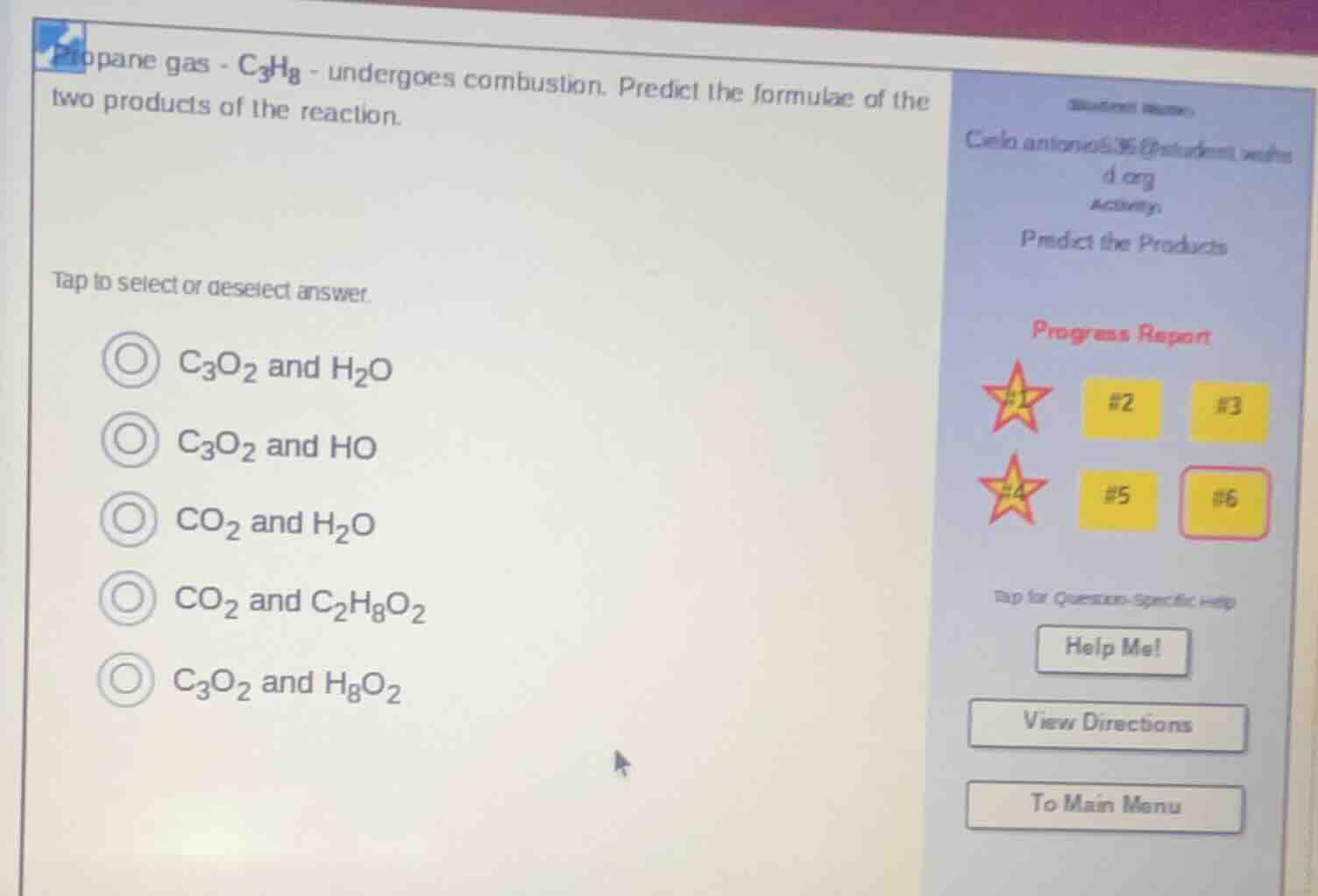
propane gas - $\ce{c3h8}$ - undergoes combustion. predict the formulae of the two products of the reaction.
tap to select or deselect answer.
$\ce{c3o2}$ and $\ce{h2o}$
$\ce{c3o2}$ and $\ce{ho}$
$\ce{co2}$ and $\ce{h2o}$
$\ce{co2}$ and $\ce{c2h8o2}$
$\ce{c3o2}$ and $\ce{h8o2}$
Combustion of hydrocarbons (like propane, \( \text{C}_3\text{H}_8 \)) in the presence of oxygen produces carbon dioxide (\( \text{CO}_2 \)) and water (\( \text{H}_2\text{O} \)). This is a standard combustion reaction where the hydrocarbon reacts with \( \text{O}_2 \) to form these products. The other options do not follow the rules of hydrocarbon combustion.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. \( \text{CO}_2 \) and \( \text{H}_2\text{O} \)