QUESTION IMAGE
Question
e⁻
v or
ps
nacl
e⁻
cu/cu²⁺
ni/ni²⁺
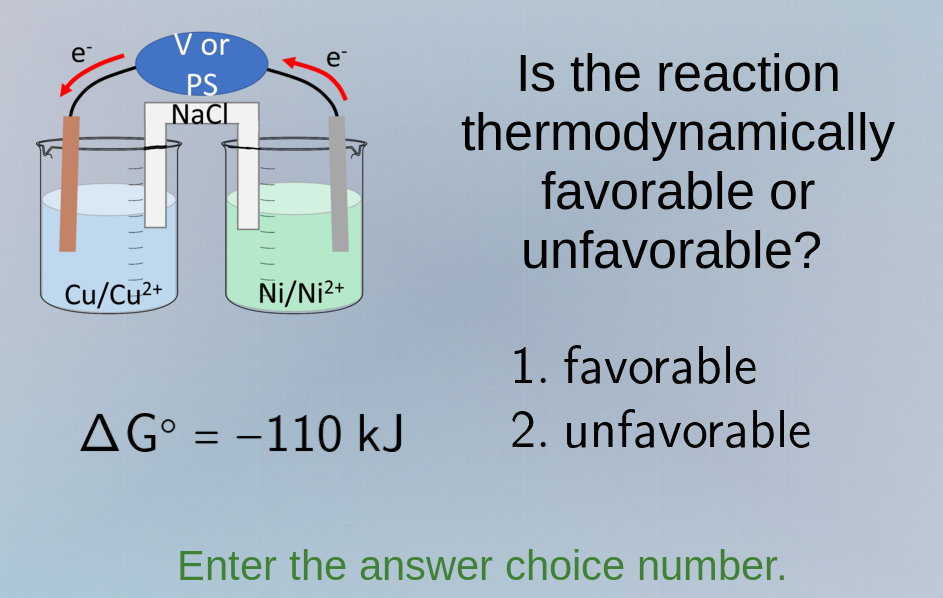
δg° = −110 kj
is the reaction thermodynamically favorable or unfavorable?
- favorable
- unfavorable
enter the answer choice number.
Brief Explanations
For a reaction, the sign of the standard Gibbs free energy change ($\Delta G^\circ$) determines thermodynamic favorability. A negative $\Delta G^\circ$ (like $\Delta G^\circ = -110\ \text{kJ}$ here) means the reaction is thermodynamically favorable (spontaneous).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1