QUESTION IMAGE
Question
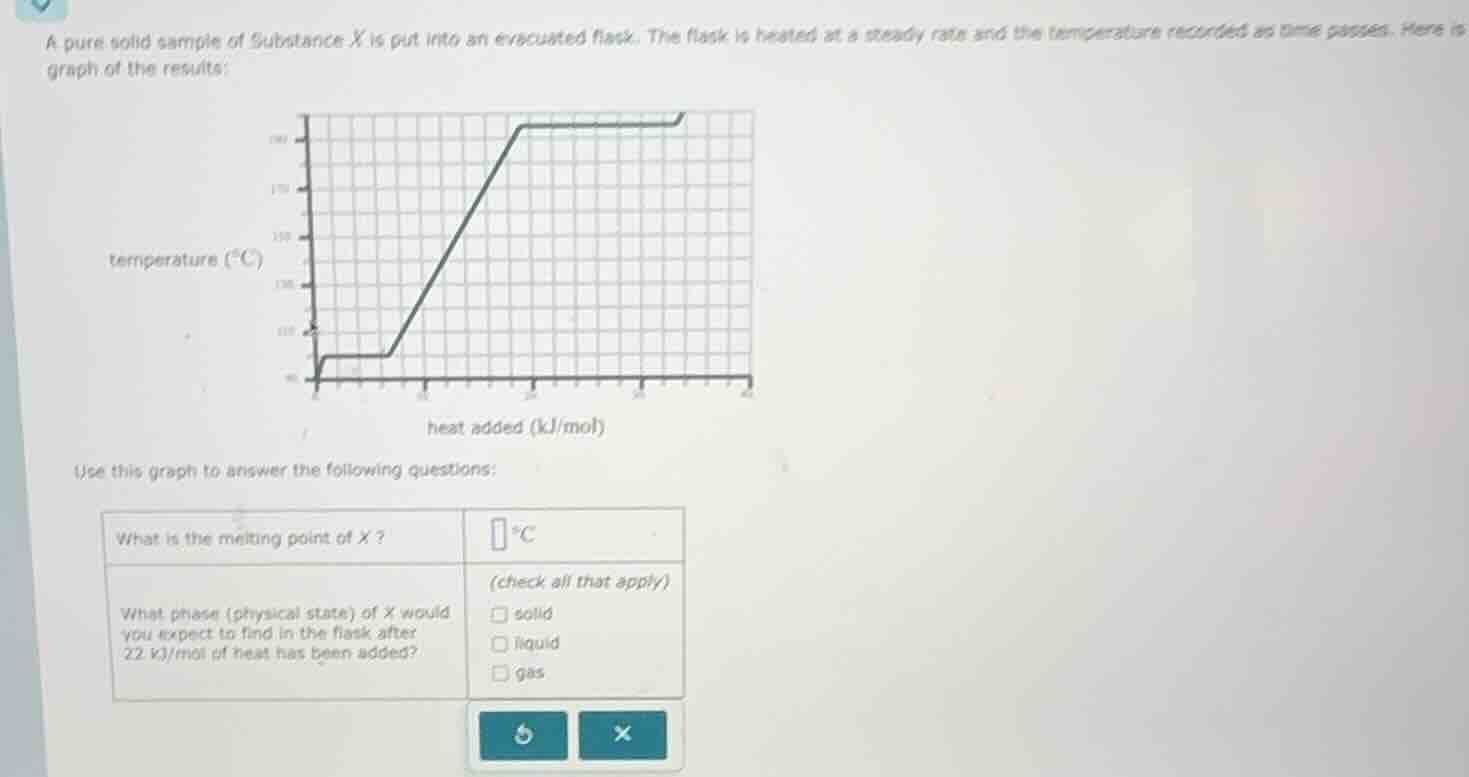
a pure solid sample of substance x is put into an evacuated flask. the flask is heated at a steady rate and the temperature recorded as time passes. here is graph of the results: use this graph to answer the following questions: what is the melting point of x? \boxed{} ^\circ c what phase (physical state) of x would you expect to find in the flask after 22 kj/mol of heat has been added? (check all that apply) \square solid \square liquid \square gas
First Question: Melting Point of X
Step1: Identify Melting Region
On a heating curve, the melting point is where the temperature is constant during melting (solid to liquid phase change). The flat segment (horizontal line) on the graph represents the phase change (melting here).
Step2: Read Temperature at Flat Segment
Looking at the graph, the flat part (melting) occurs at a temperature of 100 °C (from the y - axis, temperature scale). So the melting point is the temperature at this flat segment.
Step1: Analyze Heat Added vs. Phase Changes
First, the melting occurs at the first flat segment. Then, after melting, the temperature rises (liquid heating) until the next flat segment (boiling). The heat added of 22 kJ/mol: after melting (the first flat segment ends before 20 kJ/mol? Wait, looking at the x - axis (heat added, kJ/mol), 22 kJ/mol is after the melting (first flat) and before the boiling (second flat). Wait, no, let's re - check: the first flat is melting (solid to liquid), then temperature rises (liquid), then second flat is boiling (liquid to gas). At 22 kJ/mol, we are in the region where the temperature is rising after melting (so liquid) or before boiling? Wait, the graph: after the first flat (melting), the temperature increases (liquid heating) until the second flat (boiling). So at 22 kJ/mol, which is after the melting (first flat) and before the boiling (second flat), the substance is liquid? Wait, no, wait the second flat: when does the second flat start? The x - axis: the first flat is from 0 to around 5 kJ/mol? No, the graph: the first flat (melting) is at low heat, then temperature rises (liquid), then second flat (boiling) at higher heat. Wait, the heat added at 22 kJ/mol: looking at the x - axis, the second flat (boiling) starts around 20 kJ/mol? Wait, the graph shows that after the first flat (melting), the temperature rises (liquid) until the second flat (boiling) starts. So at 22 kJ/mol, which is in the second flat? Wait, no, the second flat is the boiling (liquid to gas). Wait, maybe I misread. Let's see: the first horizontal line (melting, solid - liquid) at around 100 °C, then temperature rises (liquid heating) to the next horizontal line (boiling, liquid - gas) at a higher temperature. The heat added at 22 kJ/mol: if the second flat (boiling) starts at around 20 kJ/mol? Wait, the x - axis: the first flat is from 0 to ~5 kJ/mol (melting), then temperature rises (liquid) until ~20 kJ/mol, then second flat (boiling) from ~20 to ~30 kJ/mol? Wait, no, the graph as per the user: the first flat (melting) at low heat, then temperature rises (liquid) to the second flat (boiling) at higher heat. So at 22 kJ/mol, we are in the second flat? No, the second flat is where temperature is constant (boiling, liquid and gas co - exist). Wait, no, when heat is added during boiling, temperature is constant (liquid to gas). So if 22 kJ/mol is in the second flat (boiling), then both liquid and gas? But the options are solid, liquid, gas. Wait, no, maybe the first flat is melting (solid - liquid), then temperature rises (liquid) until the second flat (boiling, liquid - gas). So at 22 kJ/mol, if the second flat starts at 20 kJ/mol, then at 22 kJ/mol, we are in the boiling phase (liquid and gas). But wait, the problem says "after 22 kJ/mol of heat has been added". Wait, maybe the second flat is the boiling, so at 22 kJ/mol, which is in the second flat (boiling), so the phase is liquid and gas? But the options are solid, liquid, gas (check all that apply). Wait, no, let's re - examine the graph:
- First flat: melting (solid → liquid, heat added: low, temperature 100 °C)
- Then temperature rises (liquid, heat added increasing, temperature increasing)
- Second flat: boiling (liquid → gas, heat added increasing, temperature constant)
So when heat added is 22 kJ/mol, which is in the second flat (boiling), so the substance is in liquid and gas phase? But the options are solid, liquid, gas. Wait, maybe I made a mistake. Wait, the first flat: melting (solid to liquid), then temperature rises (liquid), then second flat: boiling…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
100