QUESTION IMAGE
Question
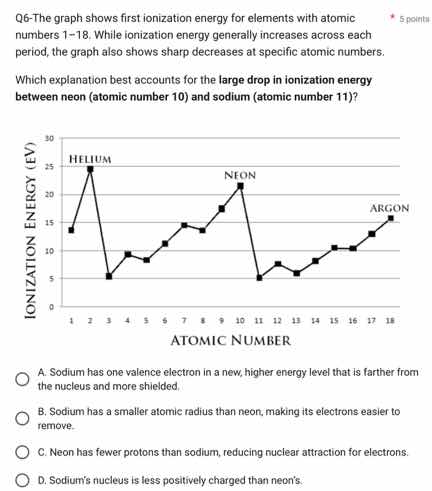
q6-the graph shows first ionization energy for elements with atomic numbers 1 - 18. while ionization energy generally increases across each period, the graph also shows sharp decreases at specific atomic numbers. which explanation best accounts for the large drop in ionization energy between neon (atomic number 10) and sodium (atomic number 11)? a. sodium has one valence electron in a new, higher energy level that is farther from the nucleus and more shielded. b. sodium has a smaller atomic radius than neon, making its electrons easier to remove. c. neon has fewer protons than sodium, reducing nuclear attraction for electrons. d. sodium’s nucleus is less positively charged than neon’s.
- Option A: Sodium (atomic number 11) has its valence electron in the 3s orbital, which is a new, higher energy level compared to neon's (atomic number 10) valence electrons in the 2p orbital. The 3s electron is farther from the nucleus and experiences more shielding from inner electrons, making it easier to remove, leading to lower ionization energy.
- Option B: Sodium has a larger atomic radius than neon, so this is incorrect.
- Option C: Neon has 10 protons, sodium has 11 protons. Neon has fewer protons, but this is not the reason for the drop (sodium has more protons but lower ionization energy due to electron configuration).
- Option D: Sodium's nucleus has 11 protons, neon's has 10 protons, so sodium's nucleus is more positively charged. This is incorrect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. Sodium has one valence electron in a new, higher energy level that is farther from the nucleus and more shielded.