QUESTION IMAGE
Question
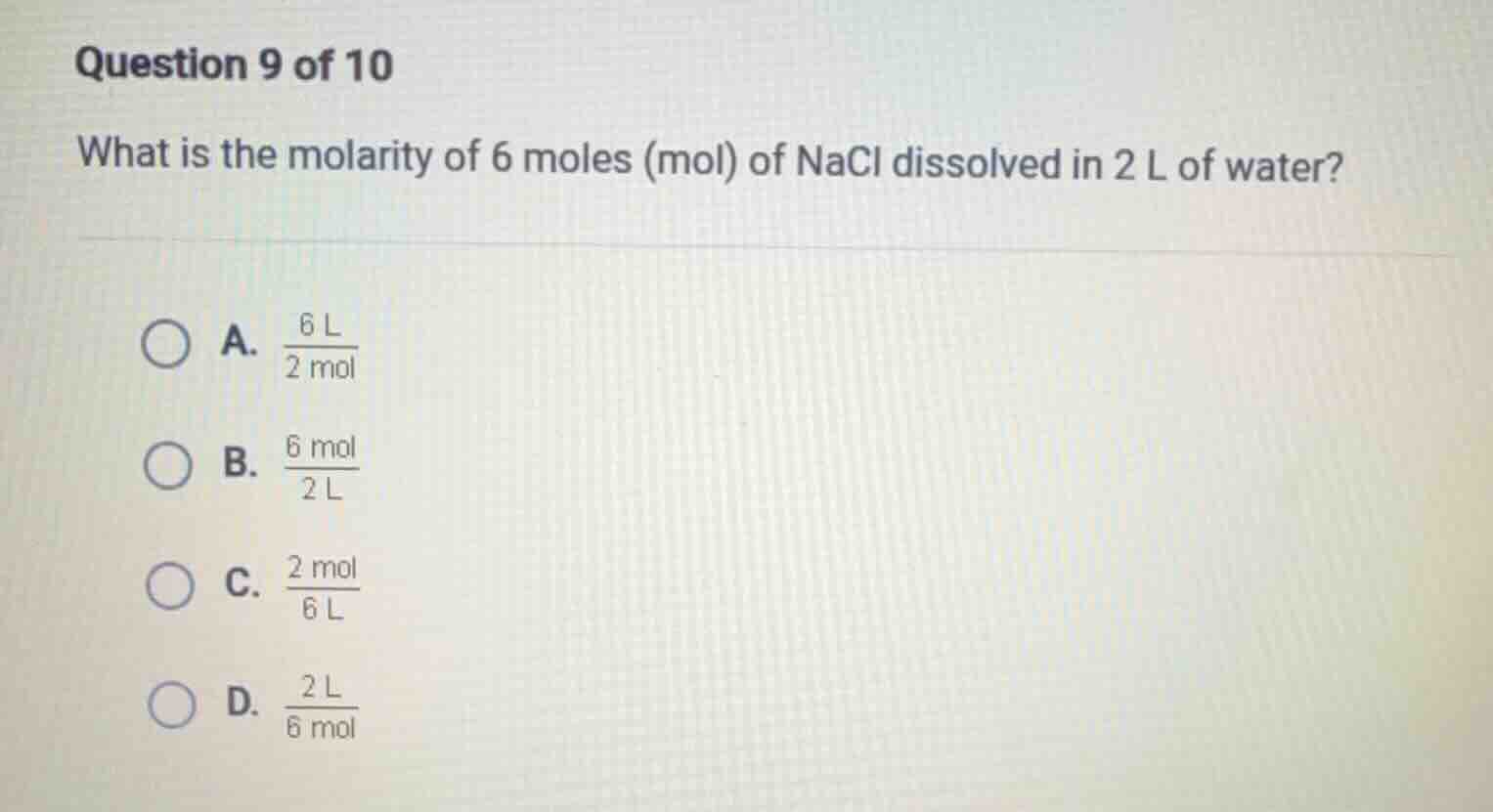
question 9 of 10
what is the molarity of 6 moles (mol) of nacl dissolved in 2 l of water?
a. \\(\frac{6\\ l}{2\\ mol}\\)
b. \\(\frac{6\\ mol}{2\\ l}\\)
c. \\(\frac{2\\ mol}{6\\ l}\\)
d. \\(\frac{2\\ l}{6\\ mol}\\)
Step1: Recall Molarity Formula
Molarity ($M$) is defined as moles of solute divided by liters of solution, so $M = \frac{\text{moles of solute}}{\text{volume of solution (in L)}}$.
Step2: Identify Values
Moles of NaCl (solute) = 6 mol, Volume of water (solution) = 2 L.
Step3: Apply Formula
Substitute values into the formula: $M = \frac{6\ \text{mol}}{2\ \text{L}}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\frac{6\ \text{mol}}{2\ \text{L}}$