QUESTION IMAGE
Question
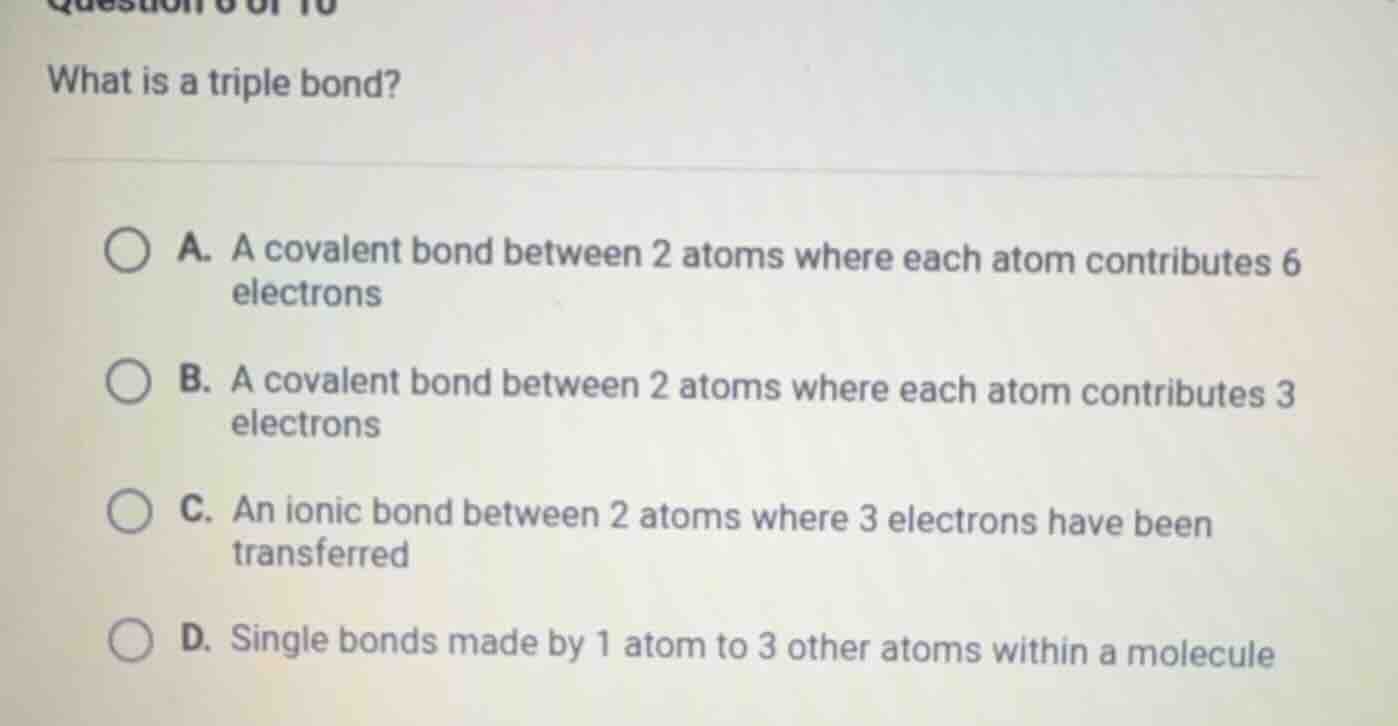
question 6 of 10
what is a triple bond?
a. a covalent bond between 2 atoms where each atom contributes 6 electrons
b. a covalent bond between 2 atoms where each atom contributes 3 electrons
c. an ionic bond between 2 atoms where 3 electrons have been transferred
d. single bonds made by 1 atom to 3 other atoms within a molecule
A triple bond is a type of covalent bond (so option C, which mentions an ionic bond, is incorrect). In a triple covalent bond, each of the two atoms contributes 3 electrons to share (a total of 6 shared electrons, 3 from each atom). Option A says each contributes 6, which is wrong. Option D describes single bonds to three atoms, not a triple bond between two atoms. So the correct option is B as it correctly describes a triple covalent bond with each atom contributing 3 electrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. A covalent bond between 2 atoms where each atom contributes 3 electrons