QUESTION IMAGE
Question
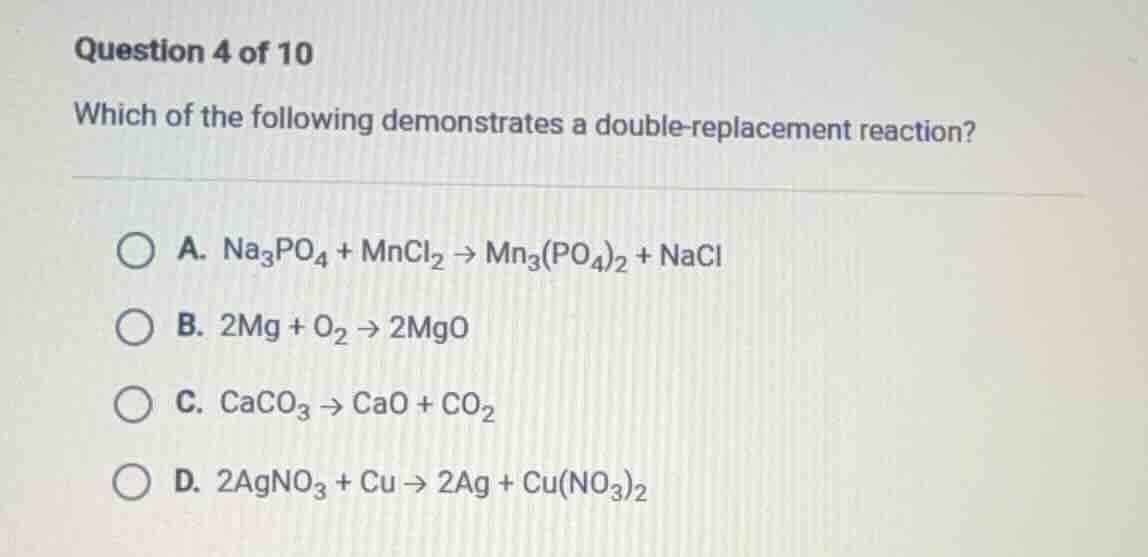
question 4 of 10
which of the following demonstrates a double - replacement reaction?
a. $\ce{na_{3}po_{4} + mncl_{2} -> mn_{3}(po_{4})_{2} + nacl}$
b. $\ce{2mg + o_{2} -> 2mgo}$
c. $\ce{caco_{3} -> cao + co_{2}}$
d. $\ce{2agno_{3} + cu -> 2ag + cu(no_{3})_{2}}$
To determine a double - replacement reaction, we recall that in a double - replacement reaction, the positive and negative ions of two ionic compounds exchange places to form two new compounds. The general form is \(AB + CD
ightarrow AD+CB\).
- Option A: In the reaction \(Na_{3}PO_{4}+MnCl_{2}
ightarrow Mn_{3}(PO_{4})_{2}+NaCl\), the sodium ion (\(Na^{+}\)) from \(Na_{3}PO_{4}\) and the manganese(II) ion (\(Mn^{2 +}\)) from \(MnCl_{2}\) exchange places with the phosphate ion (\(PO_{4}^{3-}\)) and chloride ion (\(Cl^{-}\)) respectively to form new compounds \(Mn_{3}(PO_{4})_{2}\) and \(NaCl\). This follows the pattern of a double - replacement reaction.
- Option B: The reaction \(2Mg + O_{2}
ightarrow2MgO\) is a combination (synthesis) reaction, where two substances combine to form a single product.
- Option C: The reaction \(CaCO_{3}
ightarrow CaO + CO_{2}\) is a decomposition reaction, where a single compound breaks down into two or more simpler substances.
- Option D: The reaction \(2AgNO_{3}+Cu
ightarrow2Ag + Cu(NO_{3})_{2}\) is a single - replacement reaction, where one element replaces another element in a compound.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \(Na_{3}PO_{4}+MnCl_{2}
ightarrow Mn_{3}(PO_{4})_{2}+NaCl\)