QUESTION IMAGE
Question
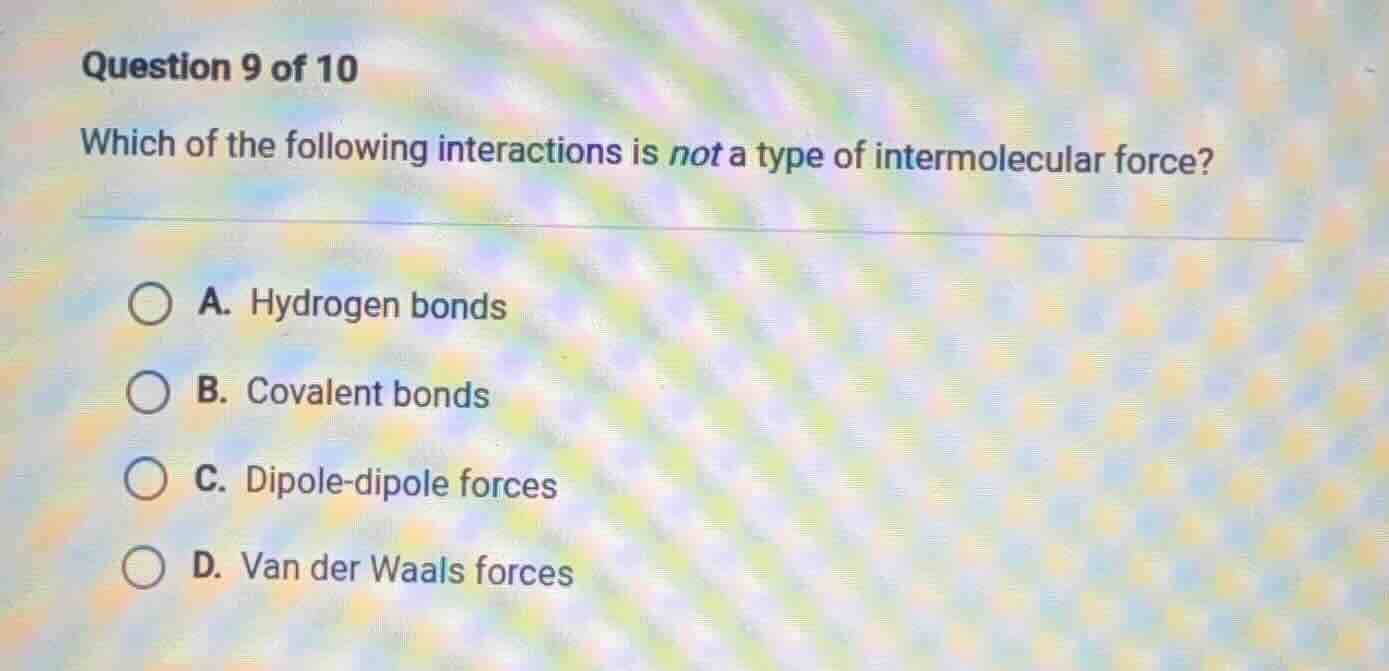
question 9 of 10
which of the following interactions is not a type of intermolecular force?
a. hydrogen bonds
b. covalent bonds
c. dipole - dipole forces
d. van der waals forces
Intermolecular forces are forces between molecules. Hydrogen bonds, dipole - dipole forces, and Van der Waals forces (including London dispersion forces) are all intermolecular forces that act between different molecules. Covalent bonds, however, are intramolecular forces, meaning they act within a molecule to hold atoms together (by sharing electrons between atoms in a molecule). So covalent bonds are not a type of intermolecular force.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. Covalent bonds