QUESTION IMAGE
Question
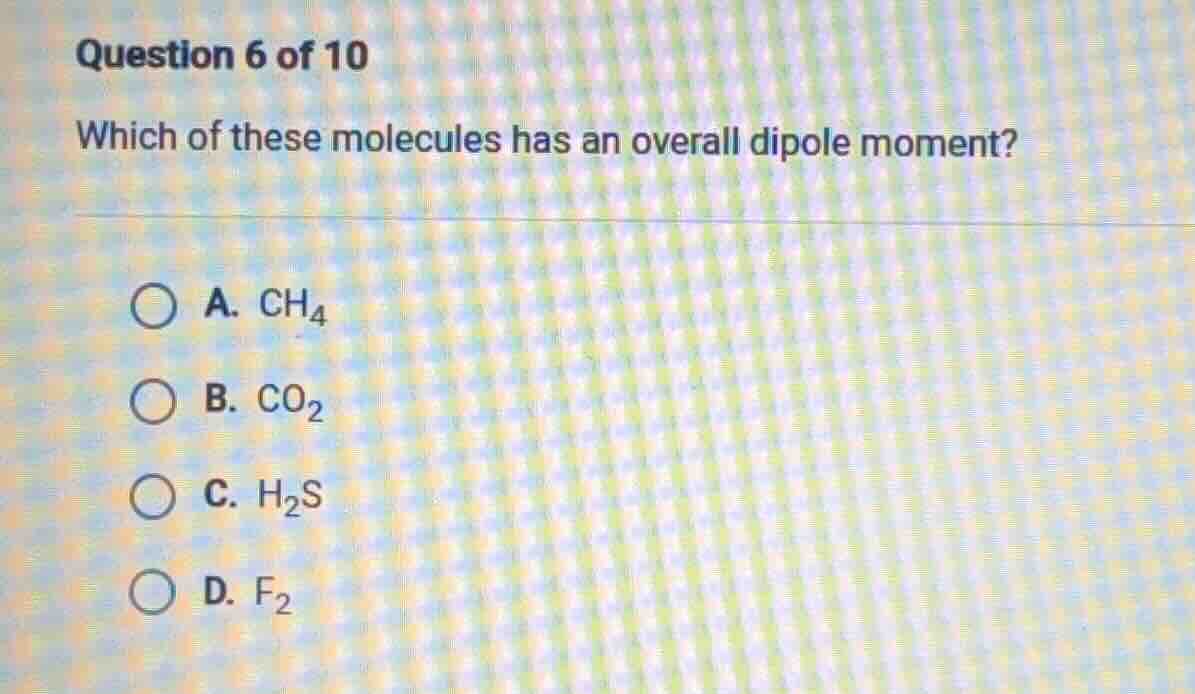
question 6 of 10
which of these molecules has an overall dipole moment?
a. ch₄
b. co₂
c. h₂s
d. f₂
Brief Explanations
To determine which molecule has an overall dipole moment, we analyze the molecular geometry and polarity:
- A. \( \text{CH}_4 \): Tetrahedral geometry, symmetric. Bond dipoles cancel, so no net dipole.
- B. \( \text{CO}_2 \): Linear geometry (\( \text{O}=\text{C}=\text{O} \)). Bond dipoles (C=O) are equal and opposite, canceling each other. No net dipole.
- C. \( \text{H}_2\text{S} \): Bent geometry (like \( \text{H}_2\text{O} \)). S-H bonds are polar, and the bent shape means bond dipoles do not cancel. Net dipole moment exists.
- D. \( \text{F}_2 \): Diatomic, nonpolar (same atom, no electronegativity difference). No dipole.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. \( \text{H}_2\text{S} \)