QUESTION IMAGE
Question
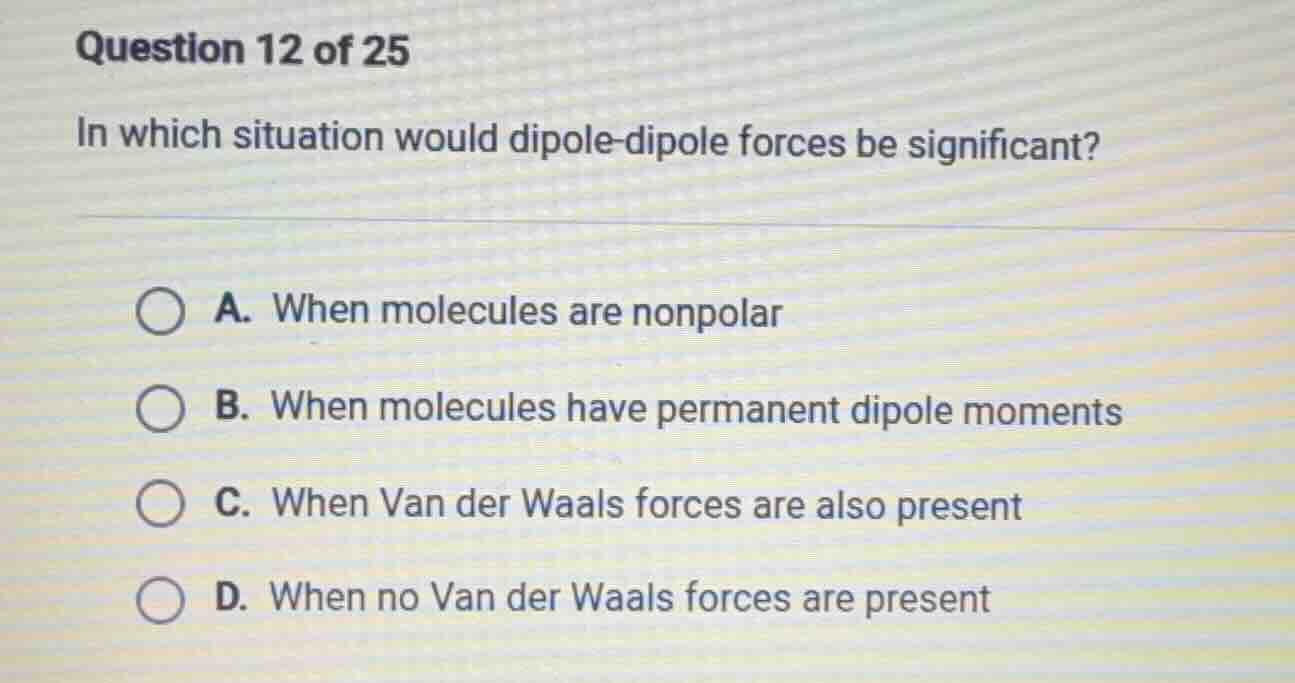
question 12 of 25
in which situation would dipole-dipole forces be significant?
a. when molecules are nonpolar
b. when molecules have permanent dipole moments
c. when van der waals forces are also present
d. when no van der waals forces are present
Dipole - dipole forces occur between molecules that have permanent dipole moments (polar molecules). Non - polar molecules do not have dipole - dipole forces (so option A is wrong). Van der Waals forces include London dispersion forces, dipole - dipole, and dipole - induced dipole forces. The presence or absence of other Van der Waals forces (like London dispersion) does not determine the significance of dipole - dipole forces. Dipole - dipole forces are a type of Van der Waals force, so the key for dipole - dipole forces to be significant is that molecules have permanent dipole moments.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. When molecules have permanent dipole moments