QUESTION IMAGE
Question
question 12 (1 point)
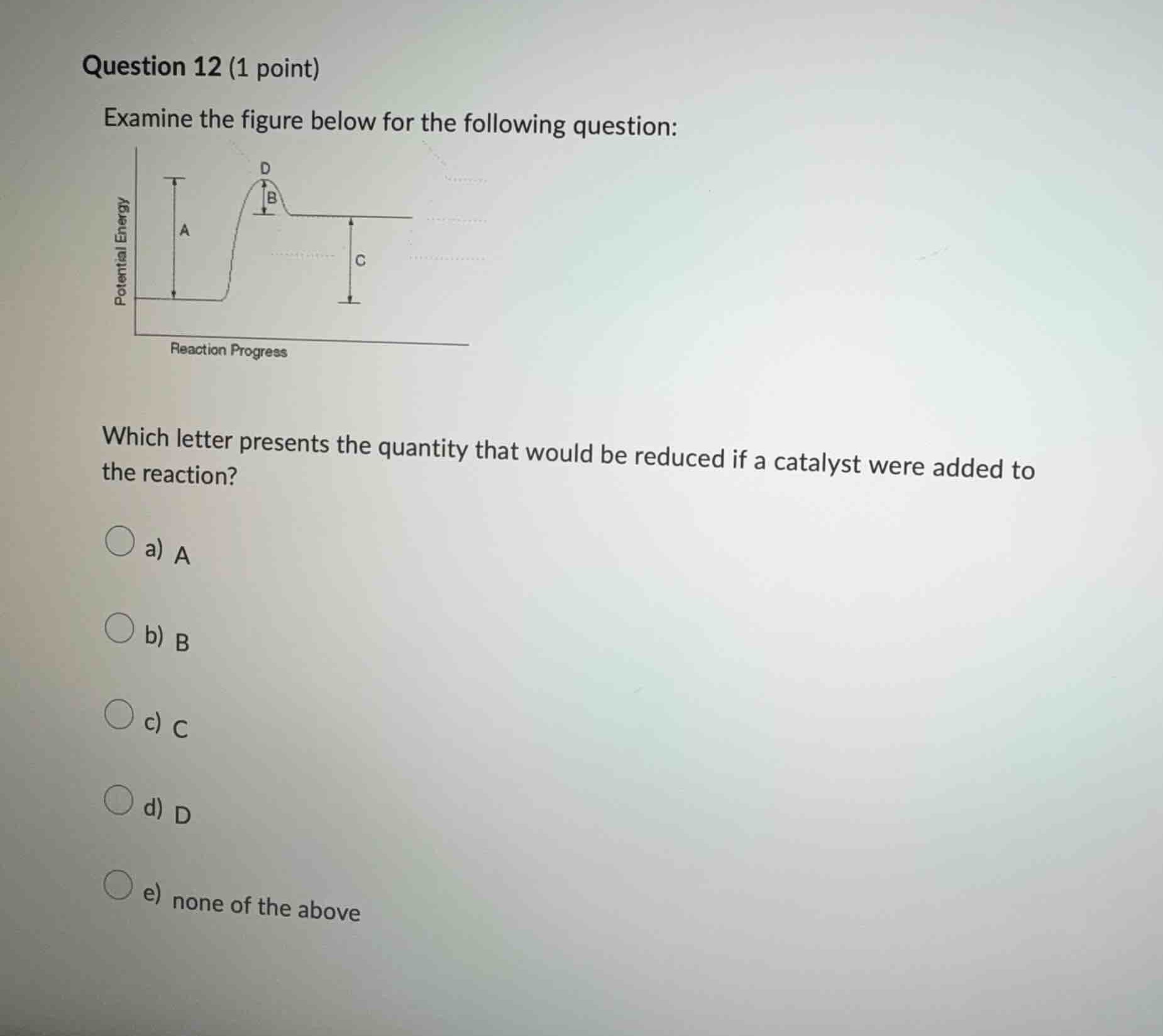
examine the figure below for the following question:
which letter presents the quantity that would be reduced if a catalyst were added to the reaction?
a) a
b) b
c) c
d) d
e) none of the above
A catalyst lowers the activation energy of a reaction. In the potential energy - reaction progress graph, the activation energy is the energy barrier that reactants must overcome to form products. The letter \( B \) represents the activation energy (the height of the energy barrier). Adding a catalyst reduces this activation energy. \( A \) represents the potential energy of reactants, \( C \) represents the potential energy difference between products and reactants (enthalpy change), and \( D \) is the peak of the energy barrier (related to activation energy but the catalyst reduces the height of this barrier which is represented by \( B \)). So the quantity reduced by a catalyst is represented by \( B \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b) B