QUESTION IMAGE
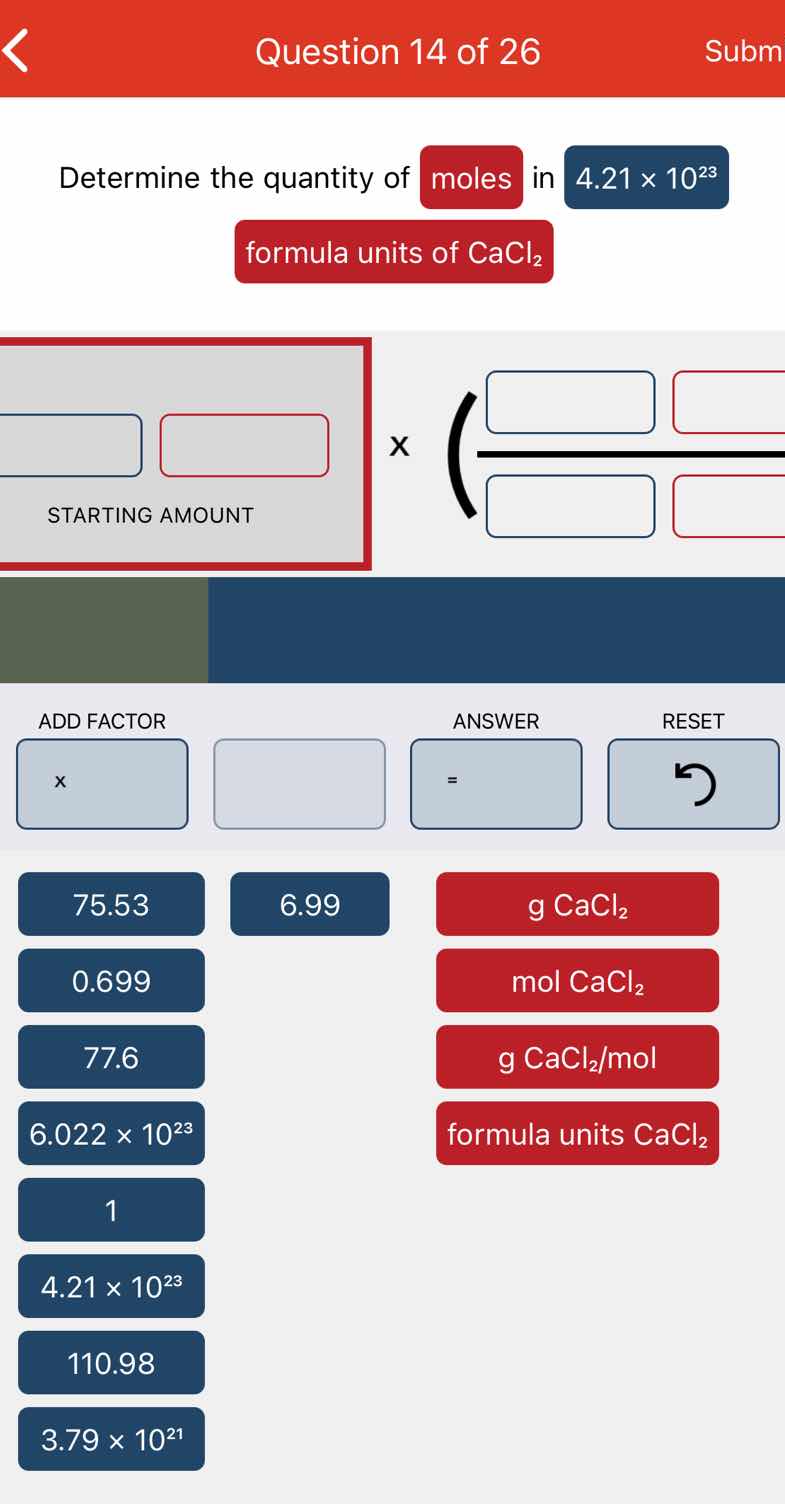
Question
question 14 of 26
determine the quantity of moles in 4.21×10²³ formula units of cacl₂
(there are some interactive elements like starting amount, add factor, answer, reset buttons and some numerical and unit options: 75.53, 6.99, 0.699, 77.6, 6.022×10²³, 1, 4.21×10²³, 110.98, g cacl₂, mol cacl₂, g cacl₂/mol, formula units cacl₂)
Step1: Identify starting amount
The given starting amount is $4.21 \times 10^{23}$ formula units of $\text{CaCl}_2$.
Step2: Use Avogadro's conversion factor
Avogadro's number is $6.022 \times 10^{23}$ formula units per mole. Set up the conversion to cancel formula units:
$$4.21 \times 10^{23} \, \text{formula units CaCl}_2 \times \frac{1 \, \text{mol CaCl}_2}{6.022 \times 10^{23} \, \text{formula units CaCl}_2}$$
Step3: Calculate the result
Perform the division:
$$\frac{4.21 \times 10^{23}}{6.022 \times 10^{23}} = 0.699$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.699 mol CaCl₂
Filled-in setup breakdown:
Starting amount: $4.21 \times 10^{23}$ formula units CaCl₂
Conversion factor: $\frac{1 \, \text{mol CaCl}_2}{6.022 \times 10^{23} \, \text{formula units CaCl}_2}$
Final calculation result: 0.699 mol CaCl₂