QUESTION IMAGE
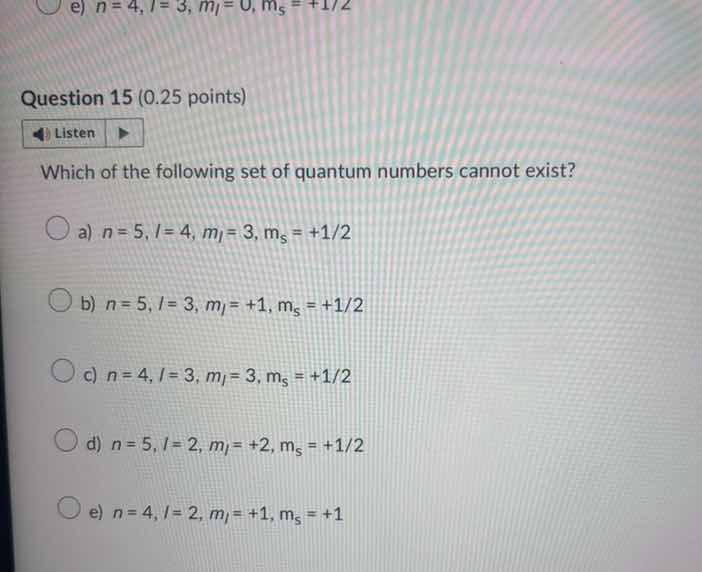
Question
question 15 (0.25 points) listen which of the following set of quantum numbers cannot exist? a) $n = 5, l = 4, m_l = 3, m_s = +1/2$ b) $n = 5, l = 3, m_l = +1, m_s = +1/2$ c) $n = 4, l = 3, m_l = 3, m_s = +1/2$ d) $n = 5, l = 2, m_l = +2, m_s = +1/2$ e) $n = 4, l = 2, m_l = +1, m_s = +1$
Step1: Recall quantum number rules
The valid ranges are:
- $n \geq 1$ (integer)
- $0 \leq l \leq n-1$ (integer)
- $-l \leq m_l \leq +l$ (integer)
- $m_s = +\frac{1}{2}$ or $-\frac{1}{2}$
Step2: Check option a
$n=5$, $l=4$ (valid, $4 \leq 5-1=4$); $m_l=3$ (valid, $-4 \leq 3 \leq 4$); $m_s=+\frac{1}{2}$ (valid).
Step3: Check option b
$n=5$, $l=3$ (valid, $3 \leq 5-1=4$); $m_l=+1$ (valid, $-3 \leq 1 \leq 3$); $m_s=+\frac{1}{2}$ (valid).
Step4: Check option c
$n=4$, $l=3$ (valid, $3 \leq 4-1=3$); $m_l=3$ (valid, $-3 \leq 3 \leq 3$); $m_s=+\frac{1}{2}$ (valid).
Step5: Check option d
$n=5$, $l=2$ (valid, $2 \leq 5-1=4$); $m_l=+2$ (valid, $-2 \leq 2 \leq 2$); $m_s=+\frac{1}{2}$ (valid).
Step6: Check option e
$n=4$, $l=2$ (valid, $2 \leq 4-1=3$); $m_l=+1$ (valid, $-2 \leq 1 \leq 2$); $m_s=+1$ (invalid, only $\pm\frac{1}{2}$ allowed).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
e) $n = 4, l = 2, m_l = +1, m_s = +1$