QUESTION IMAGE
Question
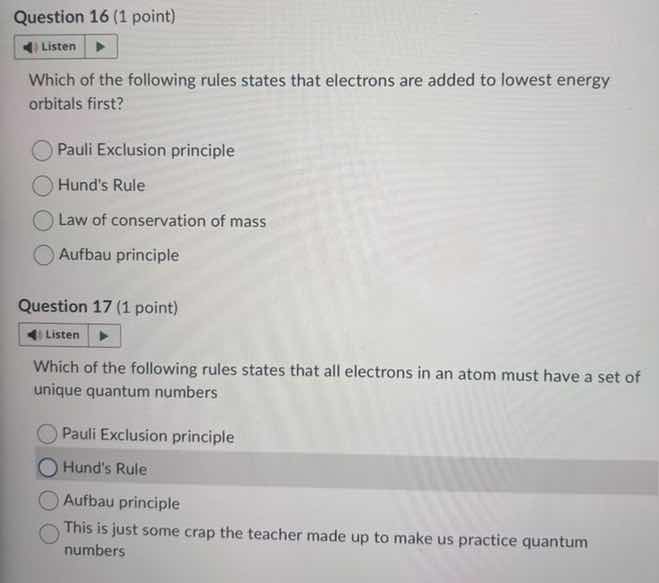
question 16 (1 point)
listen
which of the following rules states that electrons are added to lowest energy orbitals first?
pauli exclusion principle
hunds rule
law of conservation of mass
aufbau principle
question 17 (1 point)
listen
which of the following rules states that all electrons in an atom must have a set of unique quantum numbers
pauli exclusion principle
hunds rule
aufbau principle
this is just some crap the teacher made up to make us practice quantum numbers
For Question 16: The rule that describes filling lowest energy orbitals first is defined as the Aufbau principle. The Pauli Exclusion principle governs unique quantum numbers, Hund's Rule governs orbital filling with parallel spins, and the Law of Conservation of Mass relates to chemical reactions, not electron configuration.
For Question 17: The Pauli Exclusion principle dictates that no two electrons in an atom can have identical sets of quantum numbers. Hund's Rule deals with spin alignment in orbitals, and the Aufbau principle deals with filling order of orbitals. The last option is not a valid scientific rule.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 16: Aufbau principle
Question 17: Pauli Exclusion principle