QUESTION IMAGE
Question
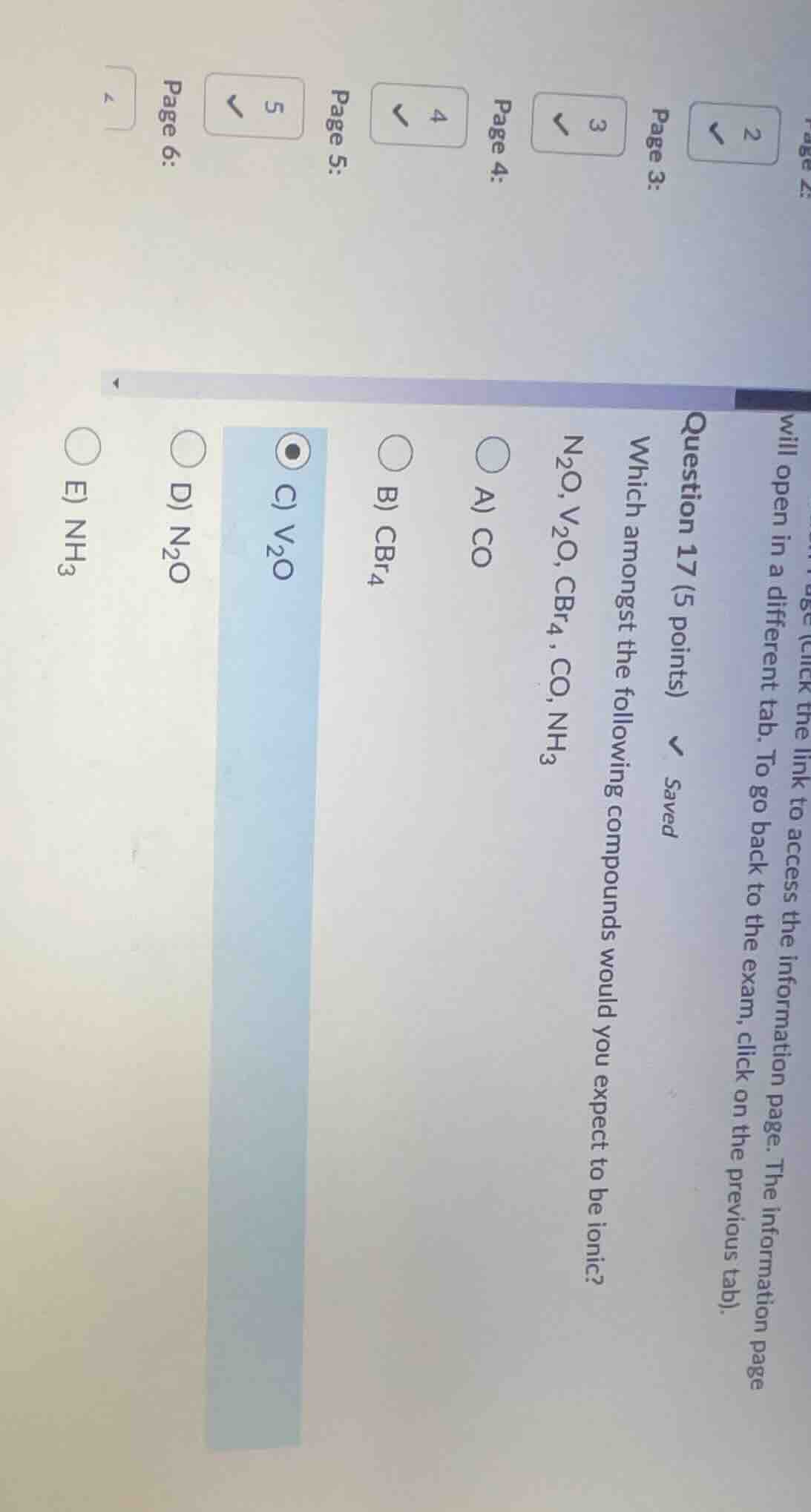
question 17 (5 points) which amongst the following compounds would you expect to be ionic? n₂o, v₂o, cbr₄, co, nh₃ a) co b) cbr₄ c) v₂o d) n₂o e) nh₃
Brief Explanations
To determine the ionic compound, we analyze the types of elements:
- Ionic compounds typically form between a metal and a non - metal.
- In option A (\(CO\)), both carbon (C) and oxygen (O) are non - metals, so it is a covalent compound.
- In option B (\(CBr_4\)), carbon (C) and bromine (Br) are non - metals, so it is a covalent compound.
- In option C (\(V_2O\)) (assuming it's \(V_2O\) or more likely \(V_2O_5\) etc., vanadium (V) is a metal and oxygen (O) is a non - metal, so it has the potential to be ionic.
- In option D (\(N_2O\)), nitrogen (N) and oxygen (O) are non - metals, so it is a covalent compound.
- In option E (\(NH_3\)), nitrogen (N) and hydrogen (H) are non - metals, so it is a covalent compound.
Among the given options, \(V_2O\) (option C) is the one that is most likely to be ionic as it consists of a metal (vanadium) and a non - metal (oxygen).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C) \(V_2O\)