QUESTION IMAGE
Question
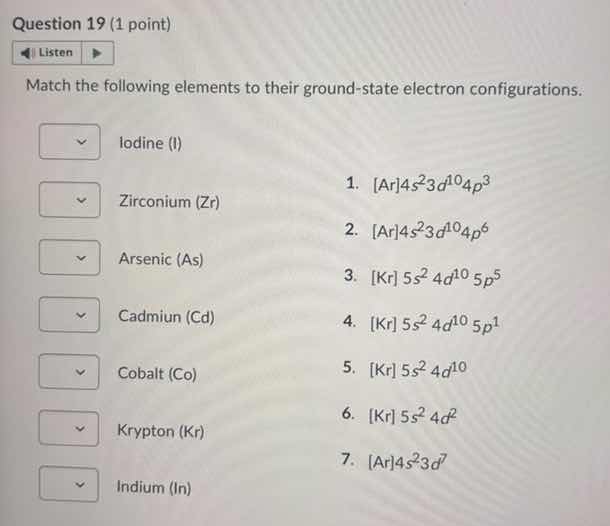
question 19 (1 point) listen match the following elements to their ground - state electron configurations. iodine (i) zirconium (zr) arsenic (as) cadmium (cd) cobalt (co) krypton (kr) indium (in) 1. ar4s²3d¹⁰4p³ 2. ar4s²3d¹⁰4p⁶ 3. kr 5s²4d¹⁰5p⁵ 4. kr 5s²4d¹⁰5p¹ 5. kr 5s²4d¹⁰ 6. kr 5s²4d² 7. ar4s²3d⁷
Brief Explanations
Each element's ground-state electron configuration is determined by its atomic number and the Aufbau principle, filling orbitals in order of increasing energy, and using noble gas shorthand for brevity.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Iodine (I): 3. $\text{[Kr]} 5s^2 4d^{10} 5p^5$
- Zirconium (Zr): 6. $\text{[Kr]} 5s^2 4d^2$
- Arsenic (As): 1. $\text{[Ar]} 4s^2 3d^{10} 4p^3$
- Cadmium (Cd): 5. $\text{[Kr]} 5s^2 4d^{10}$
- Cobalt (Co): 7. $\text{[Ar]} 4s^2 3d^7$
- Krypton (Kr): 2. $\text{[Ar]} 4s^2 3d^{10} 4p^6$
- Indium (In): 4. $\text{[Kr]} 5s^2 4d^{10} 5p^1$