QUESTION IMAGE
Question
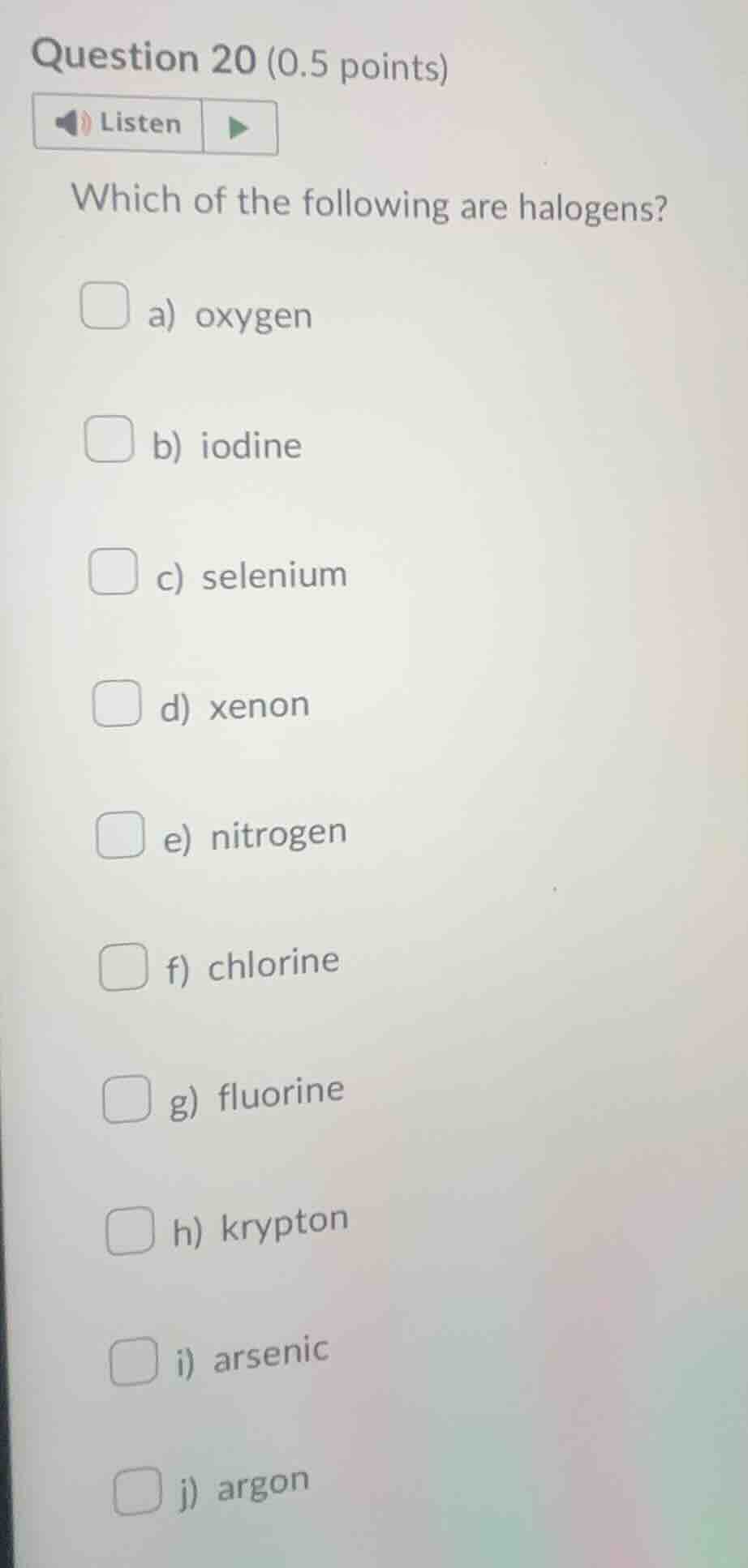
question 20 (0.5 points)listenwhich of the following are halogens?a) oxygenb) iodinec) seleniumd) xenone) nitrogenf) chlorineg) fluorineh) kryptoni) arsenicj) argon
Brief Explanations
Halogens are elements in Group 17 of the periodic table, characterized by having 7 valence electrons. We identify which options belong to this group:
- Oxygen (a) is Group 16, selenium (c) is Group 16, nitrogen (e) is Group 15, arsenic (i) is Group 15. Xenon (d), krypton (h), argon (j) are noble gases (Group 18). Iodine (b), chlorine (f), and fluorine (g) are all in Group 17, so they are halogens.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b) iodine
f) chlorine
g) fluorine