QUESTION IMAGE
Question
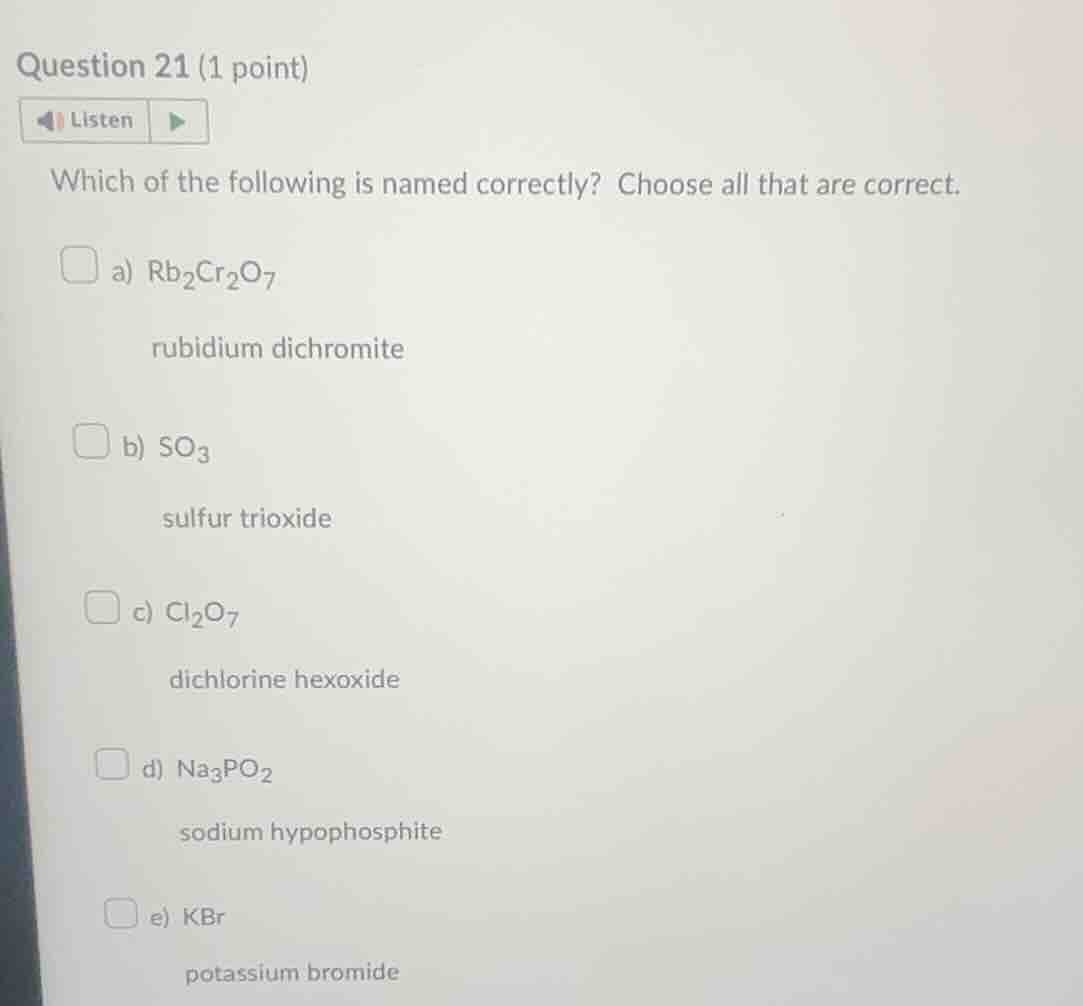
question 21 (1 point)listenwhich of the following is named correctly? choose all that are correct.a) $\text{rb}_2\text{cr}_2\text{o}_7$rubidium dichromiteb) $\text{so}_3$sulfur trioxidec) $\text{cl}_2\text{o}_7$dichlorine hexoxided) $\text{na}_3\text{po}_2$sodium hypophosphitee) kbrpotassium bromide
Step1: Analyze option a
$\text{Rb}_2\text{Cr}_2\text{O}_7$ contains the dichromate ion $\text{Cr}_2\text{O}_7^{2-}$, not dichromite. The correct name is rubidium dichromate, so this is incorrect.
Step2: Analyze option b
$\text{SO}_3$ is a covalent compound. Using prefixes, "sulfur trioxide" correctly matches 1 sulfur and 3 oxygen atoms. This is correct.
Step3: Analyze option c
$\text{Cl}_2\text{O}_7$ has 7 oxygen atoms. The correct name is dichlorine heptoxide, not hexoxide. This is incorrect.
Step4: Analyze option d
$\text{Na}_3\text{PO}_2$ contains the hypophosphite ion $\text{PO}_2^{3-}$. "Sodium hypophosphite" is the correct name for this ionic compound. This is correct.
Step5: Analyze option e
$\text{KBr}$ is an ionic compound with potassium cation $\text{K}^+$ and bromide anion $\text{Br}^-$. "Potassium bromide" is the correct name. This is correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b) $\text{SO}_3$ - sulfur trioxide
d) $\text{Na}_3\text{PO}_2$ - sodium hypophosphite
e) $\text{KBr}$ - potassium bromide