QUESTION IMAGE
Question
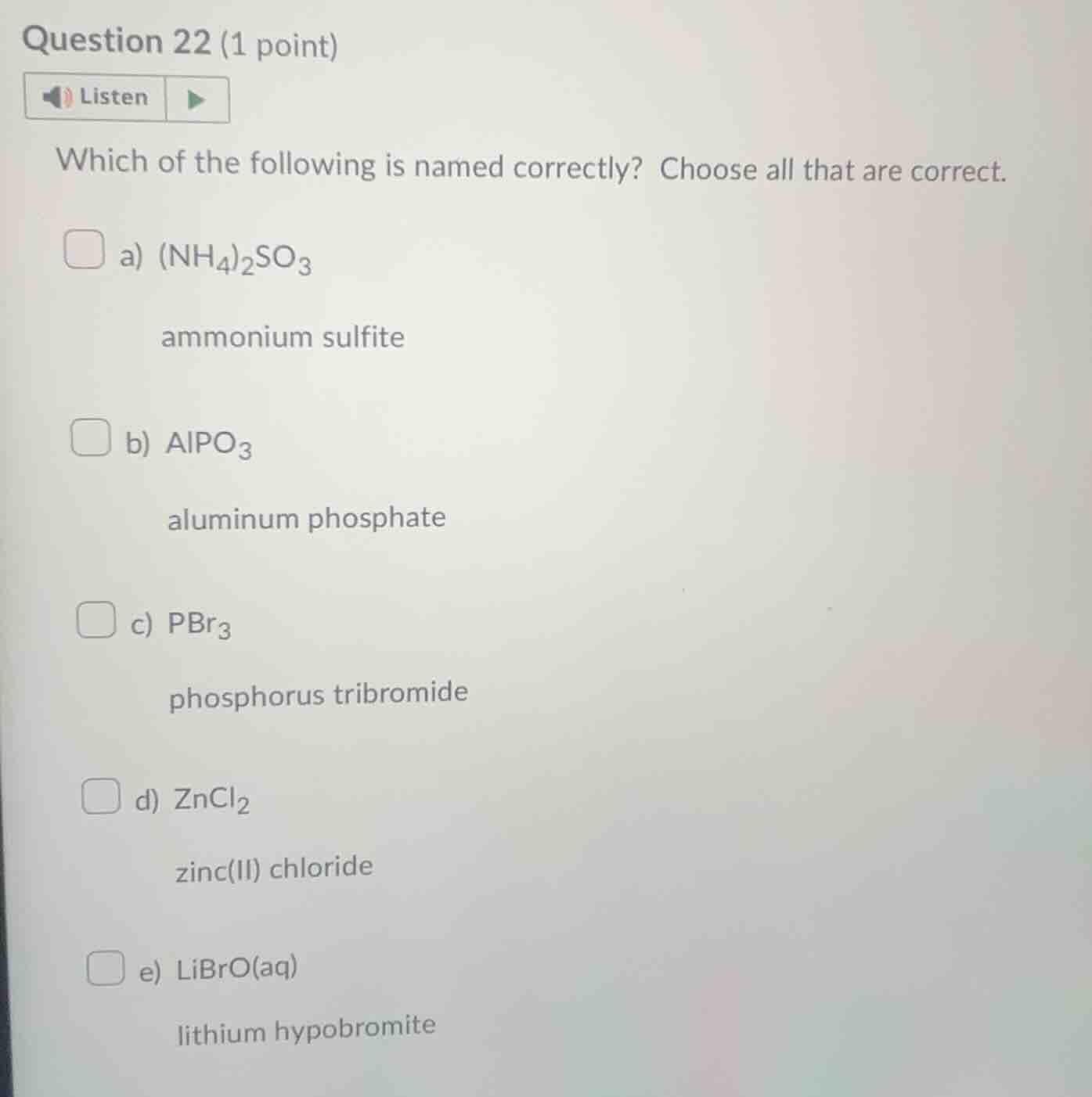
question 22 (1 point)listenwhich of the following is named correctly? choose all that are correct.a) $(nh_4)_2so_3$ammonium sulfiteb) $alpo_3$aluminum phosphatec) $pbr_3$phosphorus tribromided) $zncl_2$zinc(ii) chloridee) $libro(aq)$lithium hypobromite
Step1: Verify ionic compound a)
Ammonium ion $\text{NH}_4^+$ has +1 charge, sulfite ion $\text{SO}_3^{2-}$ has -2 charge. 2 ammonium ions balance 1 sulfite ion: $\text{(NH}_4\text{)}_2\text{SO}_3$, named ammonium sulfite. Correct.
Step2: Verify ionic compound b)
Aluminum ion $\text{Al}^{3+}$ has +3 charge, phosphate ion $\text{PO}_4^{3-}$ has -3 charge. The correct formula for aluminum phosphate is $\text{AlPO}_4$, not $\text{AlPO}_3$. Incorrect.
Step3: Verify covalent compound c)
$\text{PBr}_3$ is a covalent compound. Phosphorus is the less electronegative element, with 3 bromine atoms: named phosphorus tribromide. Correct.
Step4: Verify ionic compound d)
Zinc only forms $\text{Zn}^{2+}$ ions, so its compound is named zinc chloride (no Roman numeral needed). $\text{ZnCl}_2$ labeled zinc(II) chloride is unnecessary, so this naming is incorrect.
Step5: Verify ionic compound e)
Lithium ion $\text{Li}^+$ has +1 charge, hypobromite ion $\text{BrO}^-$ has -1 charge. Formula $\text{LiBrO(aq)}$ matches lithium hypobromite. Correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a) $\text{(NH}_4\text{)}_2\text{SO}_3$ ammonium sulfite
c) $\text{PBr}_3$ phosphorus tribromide
e) $\text{LiBrO(aq)}$ lithium hypobromite