QUESTION IMAGE
Question
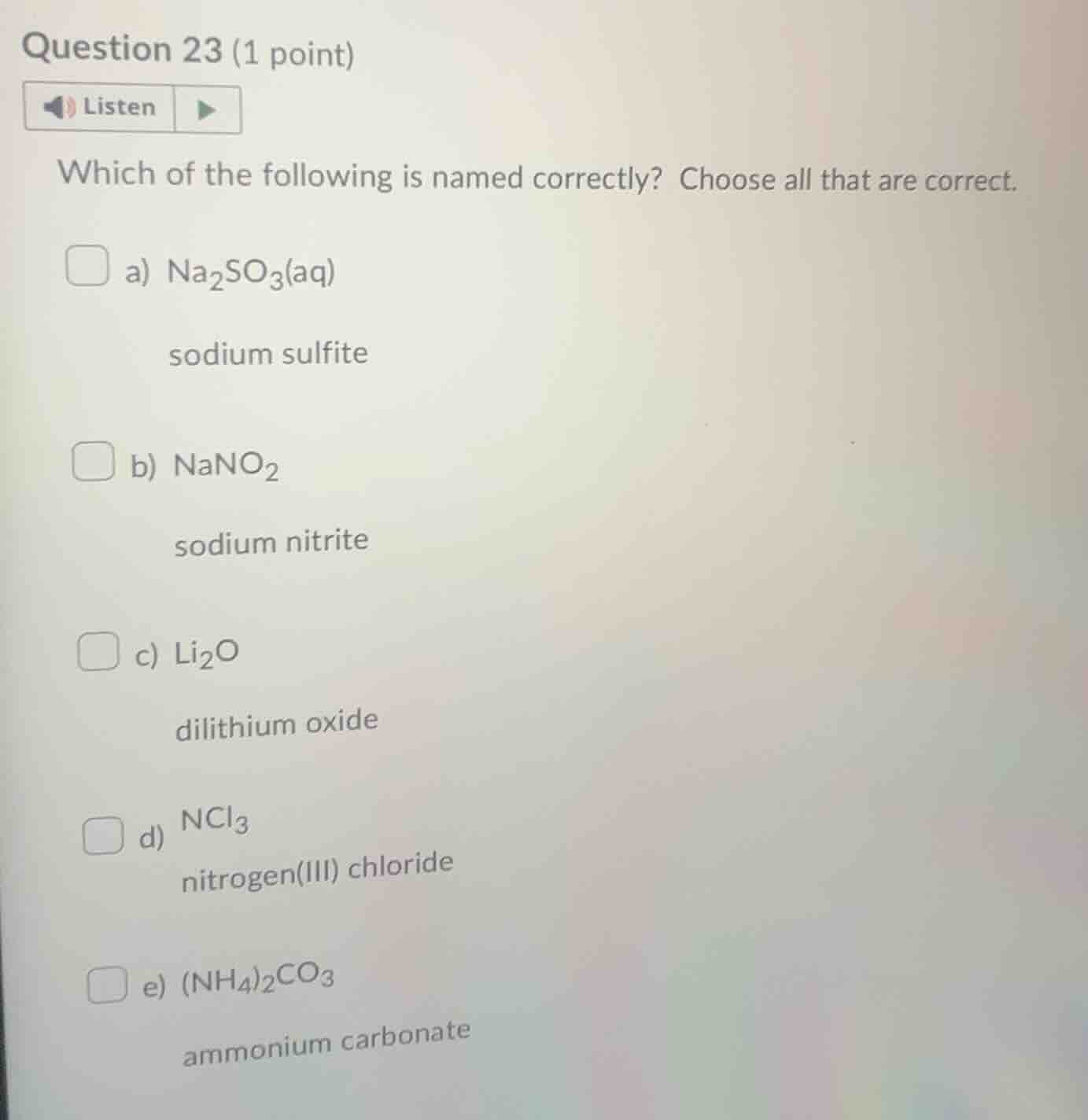
question 23 (1 point)listenwhich of the following is named correctly? choose all that are correct.a) $ce{na_{2}so_{3}(aq)}$sodium sulfiteb) $ce{nano_{2}}$sodium nitritec) $ce{li_{2}o}$dilithium oxided) $ce{ncl_{3}}$nitrogen(iii) chloridee) $ce{(nh_{4})_{2}co_{3}}$ammonium carbonate
Step1: Analyze option a
$\text{Na}_2\text{SO}_3$ has $\text{Na}^+$ (sodium cation) and $\text{SO}_3^{2-}$ (sulfite anion). The name sodium sulfite matches.
Step2: Analyze option b
$\text{NaNO}_2$ has $\text{Na}^+$ and $\text{NO}_2^-$ (nitrite anion). The name sodium nitrite matches.
Step3: Analyze option c
$\text{Li}_2\text{O}$ has $\text{Li}^+$ (lithium cation) and $\text{O}^{2-}$. For ionic compounds with group 1 metals, we do not use prefixes. The correct name is lithium oxide, not dilithium oxide.
Step4: Analyze option d
$\text{NCl}_3$ is a covalent compound. The correct name is nitrogen trichloride; using Roman numerals is for ionic compounds with variable-charge metals, which does not apply here.
Step5: Analyze option e
$(\text{NH}_4)_2\text{CO}_3$ has $\text{NH}_4^+$ (ammonium cation) and $\text{CO}_3^{2-}$ (carbonate anion). The name ammonium carbonate matches.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a) $\text{Na}_2\text{SO}_3$(aq) sodium sulfite
b) $\text{NaNO}_2$ sodium nitrite
e) $(\text{NH}_4)_2\text{CO}_3$ ammonium carbonate