QUESTION IMAGE
Question
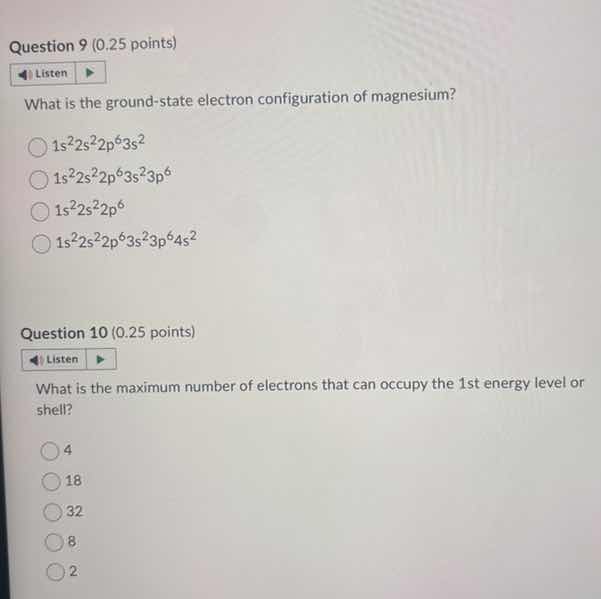
question 9 (0.25 points) listen what is the ground - state electron configuration of magnesium? 1s²2s²2p⁶3s² 1s²2s²2p⁶3s²3p⁶ 1s²2s²2p⁶ 1s²2s²2p⁶3s²3p⁶4s² question 10 (0.25 points) listen what is the maximum number of electrons that can occupy the 1st energy level or shell? 4 18 32 8 2
Question 9
Step1: Find Mg atomic number
Magnesium (Mg) has atomic number 12, so 12 electrons total.
Step2: Fill orbitals by Aufbau principle
Fill orbitals in order: $1s, 2s, 2p, 3s$.
$1s^2$ (2 electrons), $2s^2$ (2 electrons), $2p^6$ (6 electrons), remaining 2 electrons go to $3s$: $3s^2$.
Combine: $1s^22s^22p^63s^2$
Step1: Recall 1st shell orbital
The 1st energy level (n=1) only has a $1s$ orbital.
Step2: Calculate max electrons
An orbital holds max 2 electrons. $1s$ is the only orbital here, so total max electrons = 2.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$1s^22s^22p^63s^2$
---