QUESTION IMAGE
Question
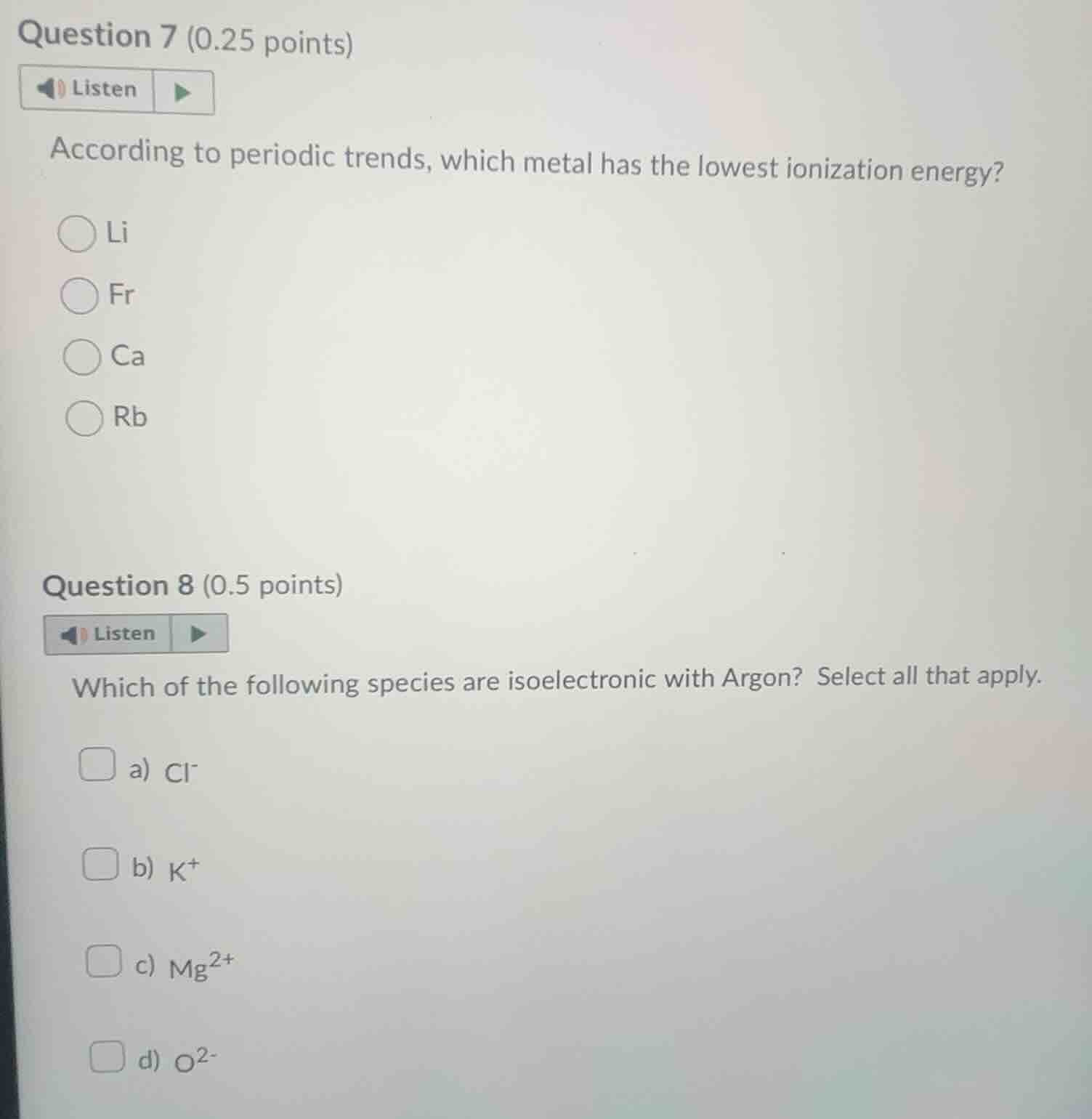
question 7 (0.25 points)listenaccording to periodic trends, which metal has the lowest ionization energy?lifrcarbquestion 8 (0.5 points)listenwhich of the following species are isoelectronic with argon? select all that apply.a) $cl^-$b) $k^+$c) $mg^{2+}$d) $o^{2-}$
For Question 7:
Ionization energy decreases down a group (as atomic radius increases, valence electrons are held less tightly) and increases across a period. Francium (Fr) is in the bottom-left of the periodic table, the lowest position in Group 1, so it has the weakest attraction to its valence electron and the lowest ionization energy among the options.
For Question 8:
Isoelectronic species have the same number of electrons. Argon (Ar) has 18 electrons.
- $\text{Cl}^-$: Chlorine has 17 electrons, gains 1 to have 18.
- $\text{K}^+$: Potassium has 19 electrons, loses 1 to have 18.
- $\text{Mg}^{2+}$: Magnesium has 12 electrons, loses 2 to have 10.
- $\text{O}^{2-}$: Oxygen has 8 electrons, gains 2 to have 10.
Only $\text{Cl}^-$ and $\text{K}^+$ match Argon's electron count.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 7: Fr
Question 8: a) $\text{Cl}^-$, b) $\text{K}^+$