QUESTION IMAGE
Question
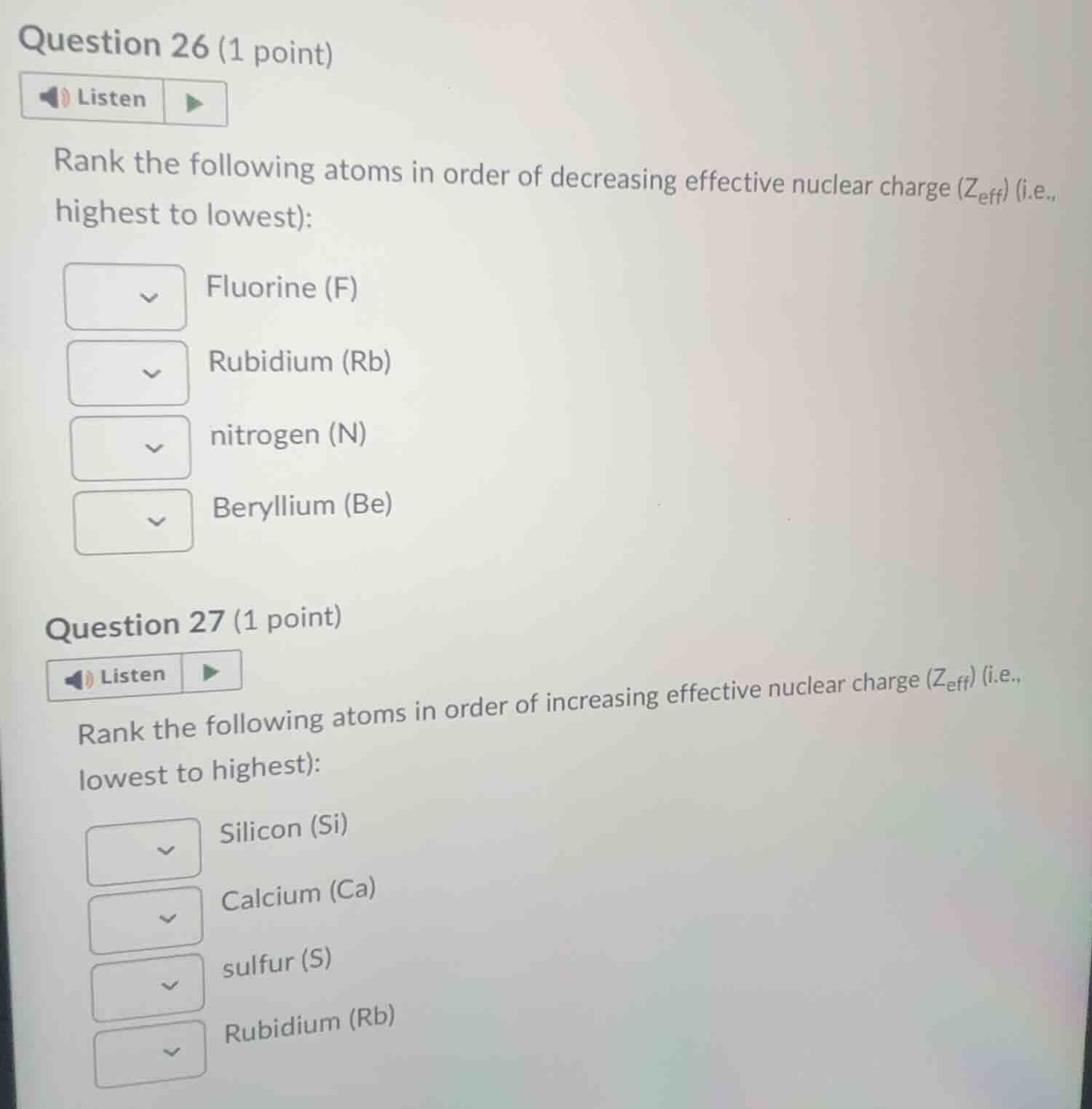
question 26 (1 point)listenrank the following atoms in order of decreasing effective nuclear charge ($z_{eff}$) (i.e., highest to lowest):fluorine (f)rubidium (rb)nitrogen (n)beryllium (be)question 27 (1 point)listenrank the following atoms in order of increasing effective nuclear charge ($z_{eff}$) (i.e., lowest to highest):silicon (si)calcium (ca)sulfur (s)rubidium (rb)
Step1: Recall $Z_{eff}$ trends
For main-group atoms:
- Across a period (left to right), $Z_{eff}$ increases (protons added, shielding from inner electrons changes little).
- Down a group (top to bottom), $Z_{eff}$ for valence electrons changes very little (protons added, but new full electron shells add significant shielding).
---
For Question 26 (Decreasing $Z_{eff}$):
Step2: List atomic numbers/periods
- F (Z=9, Period 2, Group 17)
- N (Z=7, Period 2, Group 15)
- Be (Z=4, Period 2, Group 2)
- Rb (Z=37, Period 5, Group 1)
Step3: Rank by period/group
Period 2 atoms have higher $Z_{eff}$ than Period 5 (Rb). Within Period 2, $Z_{eff}$ increases rightward.
Rank: F > N > Be > Rb
---
For Question 27 (Increasing $Z_{eff}$):
Step4: List atomic numbers/periods
- Rb (Z=37, Period 5, Group 1)
- Ca (Z=20, Period 4, Group 2)
- Si (Z=14, Period 3, Group 14)
- S (Z=16, Period 3, Group 16)
Step5: Rank by period/group
Lower period atoms have lower $Z_{eff}$ than higher periods. Within Period 3, $Z_{eff}$ increases rightward.
Rank: Rb < Ca < Si < S
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 26 (highest to lowest):
- Fluorine (F)
- nitrogen (N)
- Beryllium (Be)
- Rubidium (Rb)
Question 27 (lowest to highest):
- Rubidium (Rb)
- Calcium (Ca)
- Silicon (Si)
- sulfur (S)