QUESTION IMAGE
Question
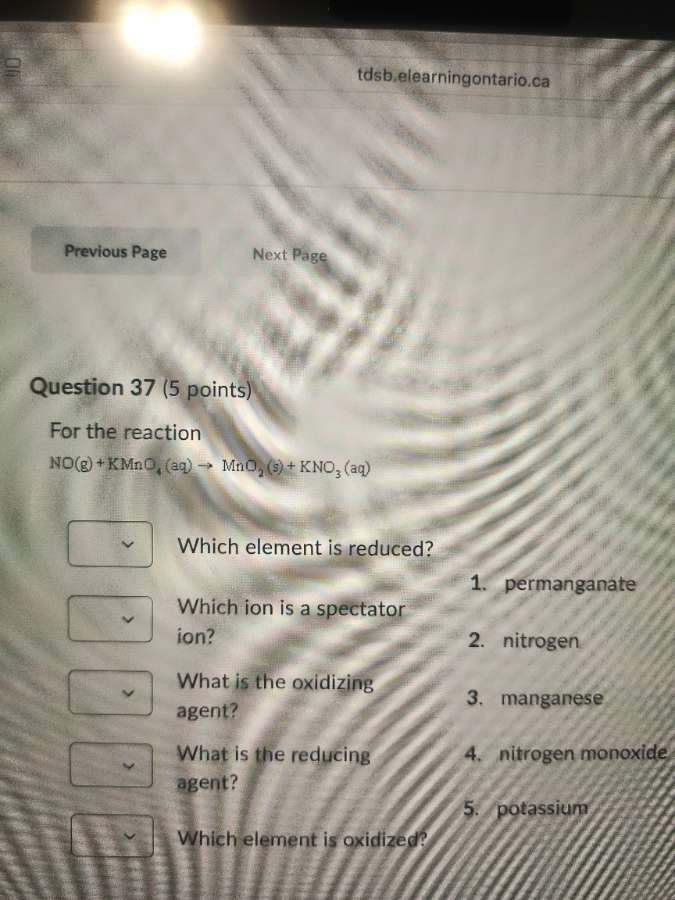
question 37 (5 points)
for the reaction
no(g) + kmno₄(aq) → mno₂(s) + kno₃(aq)
which element is reduced?
which ion is a spectator ion?
what is the oxidizing agent?
what is the reducing agent?
which element is oxidized?
- permanganate
- nitrogen
- manganese
- nitrogen monoxide
- potassium
Response
To solve this redox reaction problem, we analyze the oxidation states of each element:
Step 1: Determine oxidation states
- For \( \text{NO}(g) \): \( \text{N} \) has an oxidation state of \( +2 \), \( \text{O} \) is \( -2 \).
- For \( \text{KMnO}_4(aq) \): \( \text{K} \) is \( +1 \), \( \text{O} \) is \( -2 \), so \( \text{Mn} \) in \( \text{MnO}_4^- \) is \( +7 \) (since \( 1 + x + 4(-2) = -1 \Rightarrow x = +7 \)).
- For \( \text{MnO}_2(s) \): \( \text{O} \) is \( -2 \), so \( \text{Mn} \) is \( +4 \) (since \( x + 2(-2) = 0 \Rightarrow x = +4 \)).
- For \( \text{KNO}_3(aq) \): \( \text{K} \) is \( +1 \), \( \text{O} \) is \( -2 \), so \( \text{N} \) is \( +5 \) (since \( 1 + x + 3(-2) = 0 \Rightarrow x = +5 \)).
Step 2: Identify reduced element
- Reduction is a decrease in oxidation state. \( \text{Mn} \) goes from \( +7 \) (in \( \text{KMnO}_4 \)) to \( +4 \) (in \( \text{MnO}_2 \))—it is reduced. So the reduced element is \( \text{manganese} \) (option 3).
Step 3: Identify spectator ion
- A spectator ion does not change its oxidation state. \( \text{K}^+ \) (from \( \text{KMnO}_4 \) and \( \text{KNO}_3 \)) has an oxidation state of \( +1 \) throughout. So the spectator ion is \( \text{potassium} \) (option 5).
Step 4: Identify oxidizing agent
- The oxidizing agent is reduced (causes oxidation of another species). \( \text{KMnO}_4 \) (specifically \( \text{MnO}_4^- \), or "permanganate") is reduced (Mn goes from \( +7 \) to \( +4 \)), so the oxidizing agent is \( \text{permanganate} \) (option 1).
Step 5: Identify reducing agent
- The reducing agent is oxidized (causes reduction of another species). \( \text{NO} \) (nitrogen monoxide) is oxidized (N goes from \( +2 \) to \( +5 \)), so the reducing agent is \( \text{nitrogen monoxide} \) (option 4).
Step 6: Identify oxidized element
- Oxidation is an increase in oxidation state. \( \text{N} \) goes from \( +2 \) (in \( \text{NO} \)) to \( +5 \) (in \( \text{KNO}_3 \))—it is oxidized. So the oxidized element is \( \text{nitrogen} \) (option 2).
Final Answers:
- Which element is reduced? \( \boldsymbol{3} \) (manganese)
- Which ion is a spectator ion? \( \boldsymbol{5} \) (potassium)
- What is the oxidizing agent? \( \boldsymbol{1} \) (permanganate)
- What is the reducing agent? \( \boldsymbol{4} \) (nitrogen monoxide)
- Which element is oxidized? \( \boldsymbol{2} \) (nitrogen)
Summary:
| Question | Answer (Option) |
|---|---|
| Spectator ion | 5. potassium |
| Oxidizing agent | 1. permanganate |
| Reducing agent | 4. nitrogen monoxide |
| Oxidized element | 2. nitrogen |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this redox reaction problem, we analyze the oxidation states of each element:
Step 1: Determine oxidation states
- For \( \text{NO}(g) \): \( \text{N} \) has an oxidation state of \( +2 \), \( \text{O} \) is \( -2 \).
- For \( \text{KMnO}_4(aq) \): \( \text{K} \) is \( +1 \), \( \text{O} \) is \( -2 \), so \( \text{Mn} \) in \( \text{MnO}_4^- \) is \( +7 \) (since \( 1 + x + 4(-2) = -1 \Rightarrow x = +7 \)).
- For \( \text{MnO}_2(s) \): \( \text{O} \) is \( -2 \), so \( \text{Mn} \) is \( +4 \) (since \( x + 2(-2) = 0 \Rightarrow x = +4 \)).
- For \( \text{KNO}_3(aq) \): \( \text{K} \) is \( +1 \), \( \text{O} \) is \( -2 \), so \( \text{N} \) is \( +5 \) (since \( 1 + x + 3(-2) = 0 \Rightarrow x = +5 \)).
Step 2: Identify reduced element
- Reduction is a decrease in oxidation state. \( \text{Mn} \) goes from \( +7 \) (in \( \text{KMnO}_4 \)) to \( +4 \) (in \( \text{MnO}_2 \))—it is reduced. So the reduced element is \( \text{manganese} \) (option 3).
Step 3: Identify spectator ion
- A spectator ion does not change its oxidation state. \( \text{K}^+ \) (from \( \text{KMnO}_4 \) and \( \text{KNO}_3 \)) has an oxidation state of \( +1 \) throughout. So the spectator ion is \( \text{potassium} \) (option 5).
Step 4: Identify oxidizing agent
- The oxidizing agent is reduced (causes oxidation of another species). \( \text{KMnO}_4 \) (specifically \( \text{MnO}_4^- \), or "permanganate") is reduced (Mn goes from \( +7 \) to \( +4 \)), so the oxidizing agent is \( \text{permanganate} \) (option 1).
Step 5: Identify reducing agent
- The reducing agent is oxidized (causes reduction of another species). \( \text{NO} \) (nitrogen monoxide) is oxidized (N goes from \( +2 \) to \( +5 \)), so the reducing agent is \( \text{nitrogen monoxide} \) (option 4).
Step 6: Identify oxidized element
- Oxidation is an increase in oxidation state. \( \text{N} \) goes from \( +2 \) (in \( \text{NO} \)) to \( +5 \) (in \( \text{KNO}_3 \))—it is oxidized. So the oxidized element is \( \text{nitrogen} \) (option 2).
Final Answers:
- Which element is reduced? \( \boldsymbol{3} \) (manganese)
- Which ion is a spectator ion? \( \boldsymbol{5} \) (potassium)
- What is the oxidizing agent? \( \boldsymbol{1} \) (permanganate)
- What is the reducing agent? \( \boldsymbol{4} \) (nitrogen monoxide)
- Which element is oxidized? \( \boldsymbol{2} \) (nitrogen)
Summary:
| Question | Answer (Option) |
|---|---|
| Spectator ion | 5. potassium |
| Oxidizing agent | 1. permanganate |
| Reducing agent | 4. nitrogen monoxide |
| Oxidized element | 2. nitrogen |