QUESTION IMAGE
Question
question
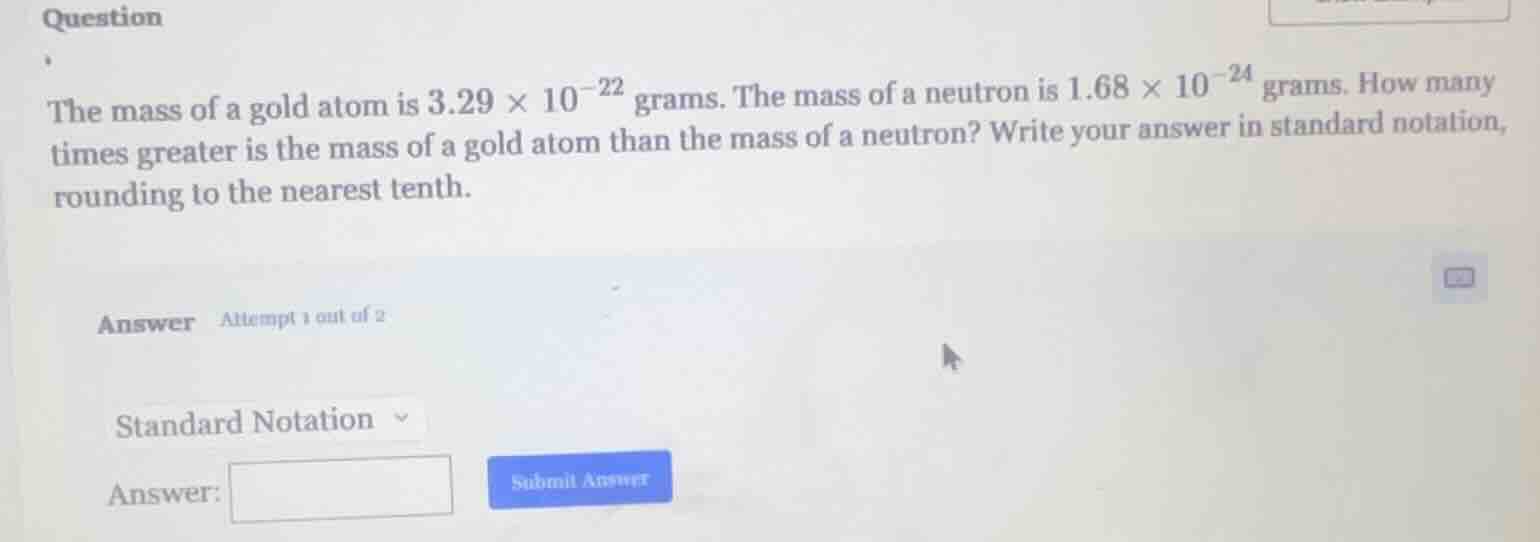
the mass of a gold atom is $3.29 \times 10^{-22}$ grams. the mass of a neutron is $1.68 \times 10^{-24}$ grams. how many times greater is the mass of a gold atom than the mass of a neutron? write your answer in standard notation, rounding to the nearest tenth.
answer attempt 1 out of 2
standard notation
answer:
submit answer
Step1: Divide the mass of gold atom by the mass of neutron
To find how many times greater the mass of a gold atom is than the mass of a neutron, we divide the mass of the gold atom by the mass of the neutron. So we have the expression: $\frac{3.29\times 10^{-22}}{1.68\times 10^{-24}}$
Step2: Use the rule of exponents for division ($\frac{a\times10^{m}}{b\times10^{n}}=\frac{a}{b}\times10^{m - n}$)
First, divide the coefficients: $\frac{3.29}{1.68}\approx1.9583$
Then, divide the powers of 10: $10^{-22-(-24)} = 10^{-22 + 24}=10^{2}$
Multiply the two results together: $1.9583\times10^{2}=195.83$
Step3: Round to the nearest tenth
Rounding 195.83 to the nearest tenth gives 195.8
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
195.8