QUESTION IMAGE
Question
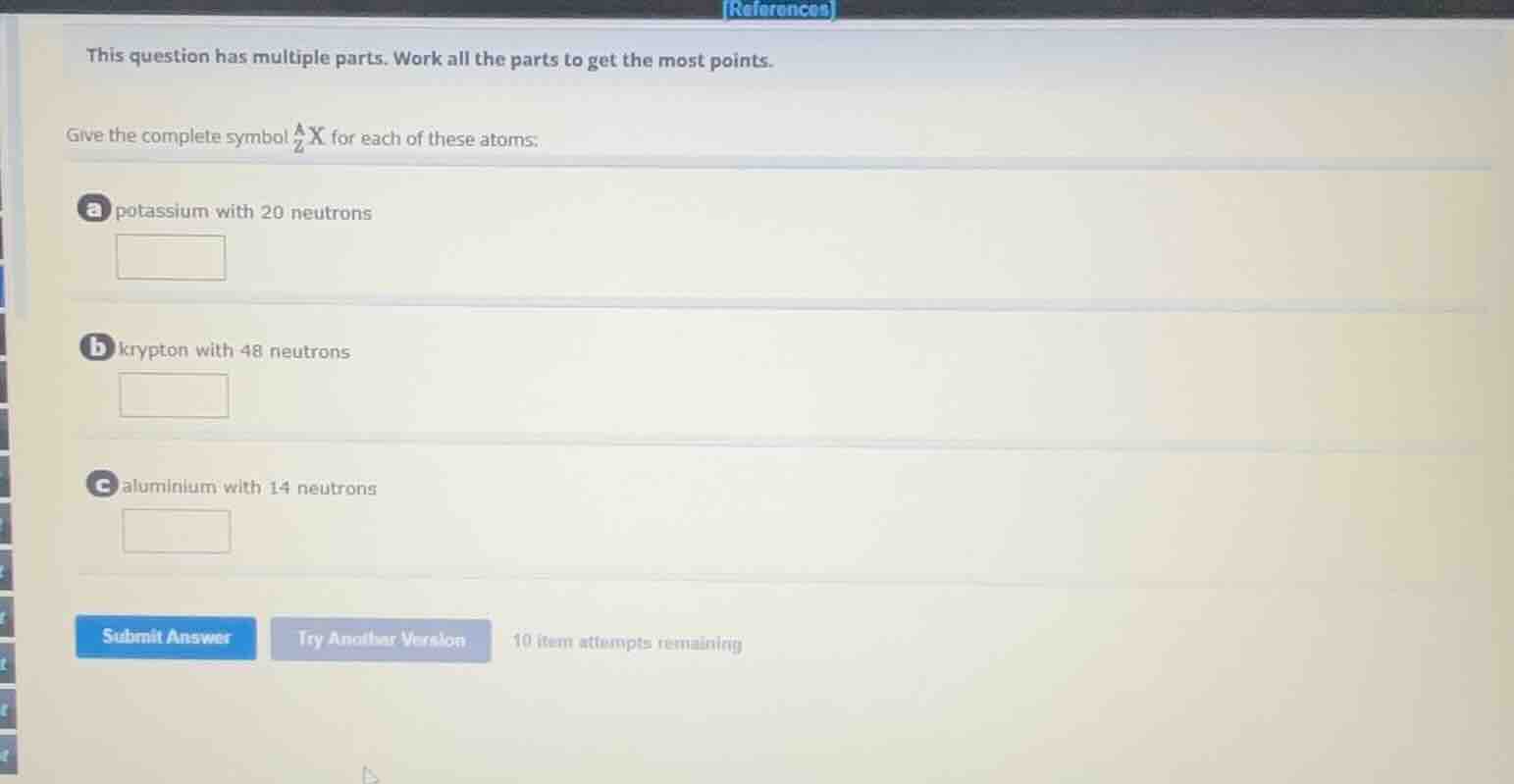
this question has multiple parts. work all the parts to get the most points. give the complete symbol $_{z}^{a}\text{x}$ for each of these atoms: a potassium with 20 neutrons b krypton with 48 neutrons c aluminium with 14 neutrons
Part (a)
Step1: Find atomic number of potassium
Potassium (K) has atomic number \( Z = 19 \).
Step2: Calculate mass number
Mass number \( A = \text{number of protons} + \text{number of neutrons} \). Number of protons = atomic number = 19, neutrons = 20. So \( A = 19 + 20 = 39 \).
Step3: Write the symbol
The symbol is \( \ce{_{19}^{39}K} \).
Part (b)
Step1: Find atomic number of krypton
Krypton (Kr) has atomic number \( Z = 36 \).
Step2: Calculate mass number
\( A = 36 + 48 = 84 \).
Step3: Write the symbol
The symbol is \( \ce{_{36}^{84}Kr} \).
Part (c)
Step1: Find atomic number of aluminium
Aluminium (Al) has atomic number \( Z = 13 \).
Step2: Calculate mass number
\( A = 13 + 14 = 27 \).
Step3: Write the symbol
The symbol is \( \ce{_{13}^{27}Al} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
a. \( \boldsymbol{\ce{_{19}^{39}K}} \)
b. \( \boldsymbol{\ce{_{36}^{84}Kr}} \)
c. \( \boldsymbol{\ce{_{13}^{27}Al}} \)