QUESTION IMAGE
Question
question 5 (1 point)
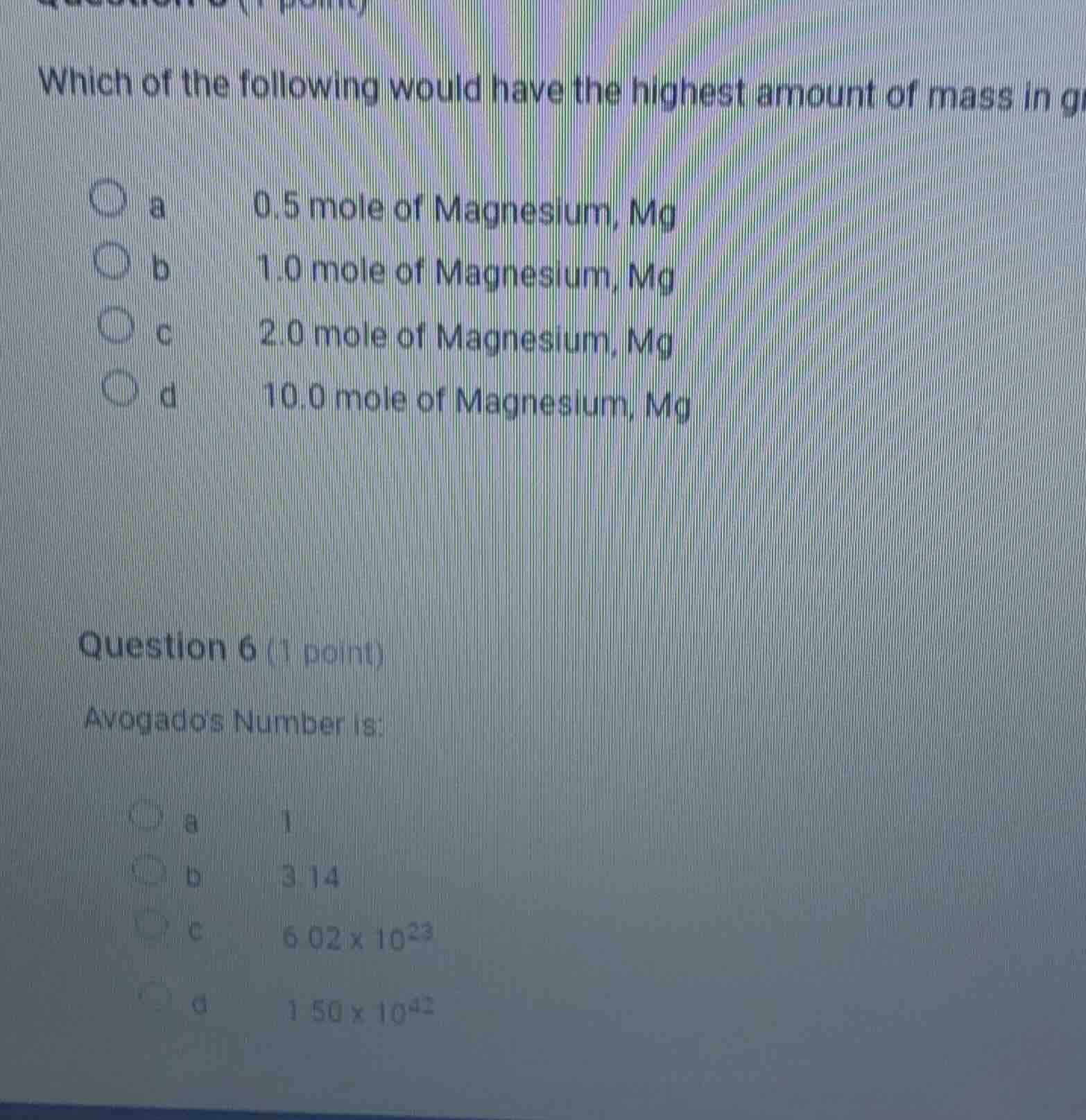
which of the following would have the highest amount of mass in g
a 0.5 mole of magnesium, mg
b 1.0 mole of magnesium, mg
c 2.0 mole of magnesium, mg
d 10.0 mole of magnesium, mg
question 6 (1 point)
avogadros number is:
a 1
b 3.14
c 6.02 x 10²³
d 1.50 x 10⁴²
Question 5 (Mass of Magnesium)
Step1: Recall the formula for mass from moles
The formula to calculate mass from moles is \( \text{Mass} = \text{Moles} \times \text{Molar Mass} \). The molar mass of Magnesium (Mg) is a constant (approximately \( 24.305 \, \text{g/mol} \)).
Step2: Analyze the relationship between moles and mass
Since the molar mass of Mg is constant, the mass is directly proportional to the number of moles (\( \text{Mass} \propto \text{Moles} \)). So, the more moles of Mg, the higher the mass.
Step3: Compare the number of moles in each option
- Option a: \( 0.5 \, \text{mole} \)
- Option b: \( 1.0 \, \text{mole} \)
- Option c: \( 2.0 \, \text{mole} \)
- Option d: \( 10.0 \, \text{mole} \)
Among these, \( 10.0 \, \text{mole} \) is the largest number of moles. So, \( 10.0 \, \text{mole} \) of Mg will have the highest mass.
Avogadro's number is defined as the number of particles (atoms, molecules, ions, etc.) in one mole of a substance. By definition, Avogadro's number is approximately \( 6.02 \times 10^{23} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d. 10.0 mole of Magnesium, Mg