QUESTION IMAGE
Question
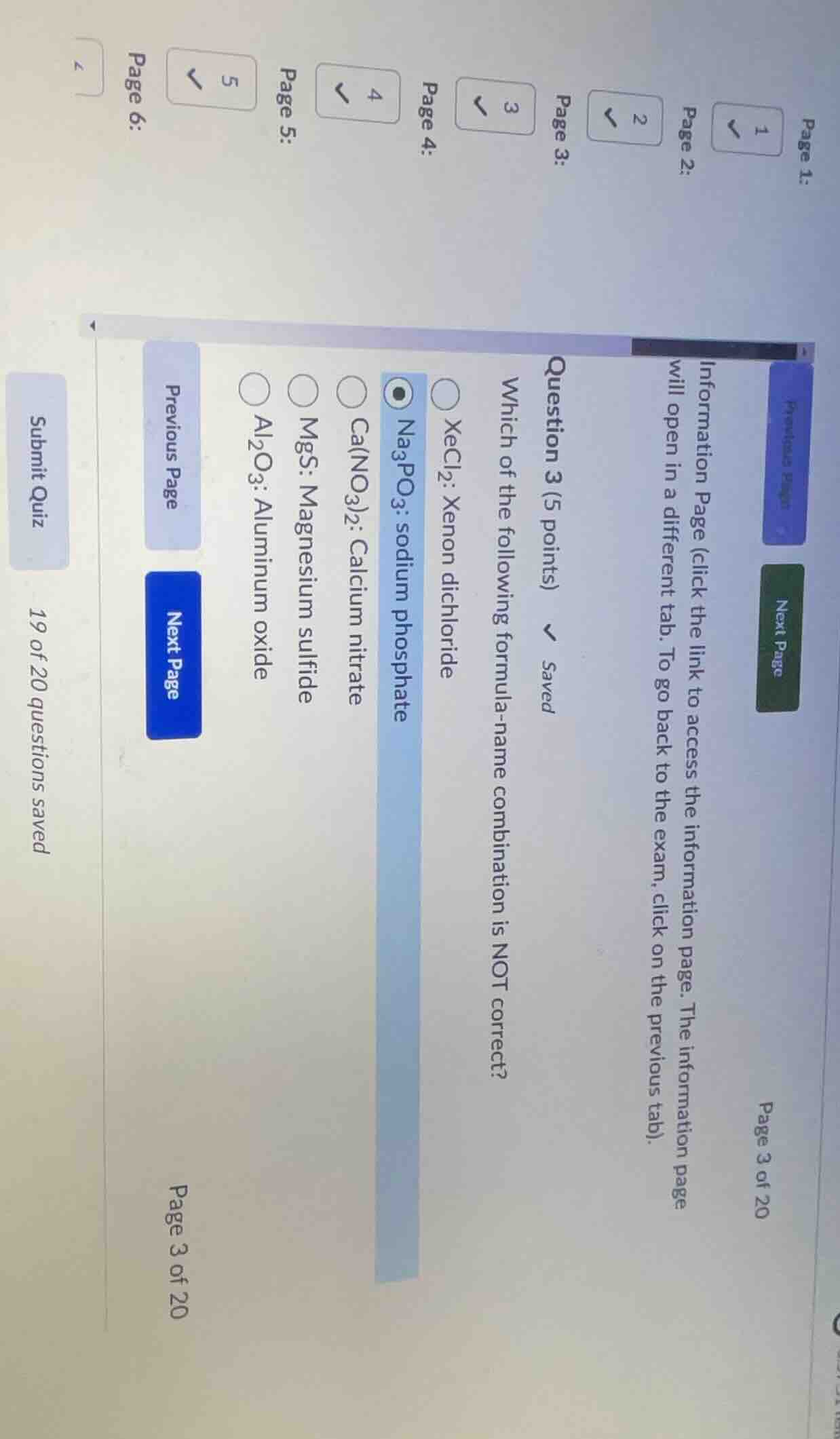
question 3 (5 points) \\(\boldsymbol{\downarrow}\\) saved which of the following formula-name combination is not correct? \\(\circ\\) xecl₂: xenon dichloride \\(\boldsymbol{\bullet}\\) na₃po₃: sodium phosphate \\(\circ\\) ca(no₃)₂: calcium nitrate \\(\circ\\) mgs: magnesium sulfide \\(\circ\\) al₂o₃: aluminum oxide
Brief Explanations
To determine the incorrect formula - name combination, we analyze each option:
- For \(XeCl_2\): Xenon is a noble gas, and when naming binary compounds with non - metals, we use prefixes. The prefix "di - " is used for 2 chlorine atoms, so "xenon dichloride" is correct.
- For \(Na_3PO_3\): The correct formula for sodium phosphate is \(Na_3PO_4\) (since the phosphate ion is \(PO_4^{3 - }\)). The ion in \(Na_3PO_3\) is \(PO_3^{3 - }\) (phosphite ion), so the correct name should be sodium phosphite, not sodium phosphate.
- For \(Ca(NO_3)_2\): Calcium has a \(+2\) charge, and the nitrate ion (\(NO_3^-\)) has a \(- 1\) charge. To balance the charges, we need 2 nitrate ions for 1 calcium ion, and the name "calcium nitrate" is correct.
- For \(MgS\): Magnesium has a \(+2\) charge, and the sulfide ion (\(S^{2 - }\)) has a \(-2\) charge. The formula \(MgS\) is correct, and the name "magnesium sulfide" is also correct.
- For \(Al_2O_3\): Aluminum has a \(+3\) charge, and the oxide ion (\(O^{2 - }\)) has a \(-2\) charge. Using the criss - cross method, we get \(Al_2O_3\), and the name "aluminum oxide" is correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The incorrect formula - name combination is \(Na_3PO_3\): sodium phosphate (the option with this combination).