QUESTION IMAGE
Question
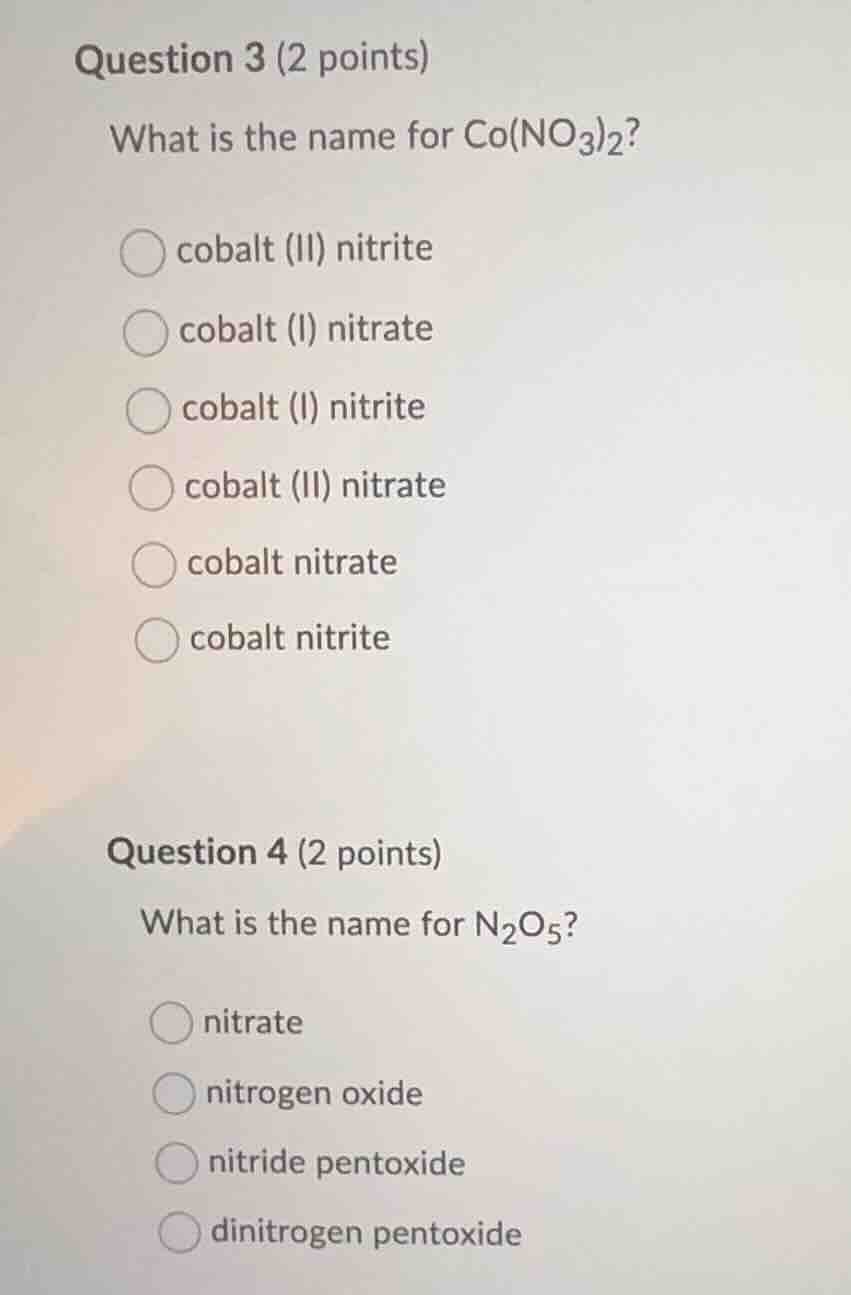
question 3 (2 points)
what is the name for co(no₃)₂?
○ cobalt (ii) nitrite
○ cobalt (i) nitrate
○ cobalt (i) nitrite
○ cobalt (ii) nitrate
○ cobalt nitrate
○ cobalt nitrite
question 4 (2 points)
what is the name for n₂o₅?
○ nitrate
○ nitrogen oxide
○ nitride pentoxide
○ dinitrogen pentoxide
Question 3
To name \( \text{Co(NO}_3\text{)}_2 \), first identify the ions. The nitrate ion is \( \text{NO}_3^- \), and there are 2 of them, so the total negative charge is \( 2\times(-1) = -2 \). Thus, cobalt must have a \( +2 \) charge (cobalt (II)). The anion is nitrate (\( \text{NO}_3^- \), not nitrite \( \text{NO}_2^- \)). So the name is cobalt (II) nitrate.
For \( \text{N}_2\text{O}_5 \), we use prefixes for covalent compounds. "Di-" for 2 nitrogen atoms and "penta-" for 5 oxygen atoms. The compound is covalent (non - metal + non - metal), so we name it using prefixes. So it is dinitrogen pentoxide.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. cobalt (II) nitrate