QUESTION IMAGE
Question
question 3 1.5 pts
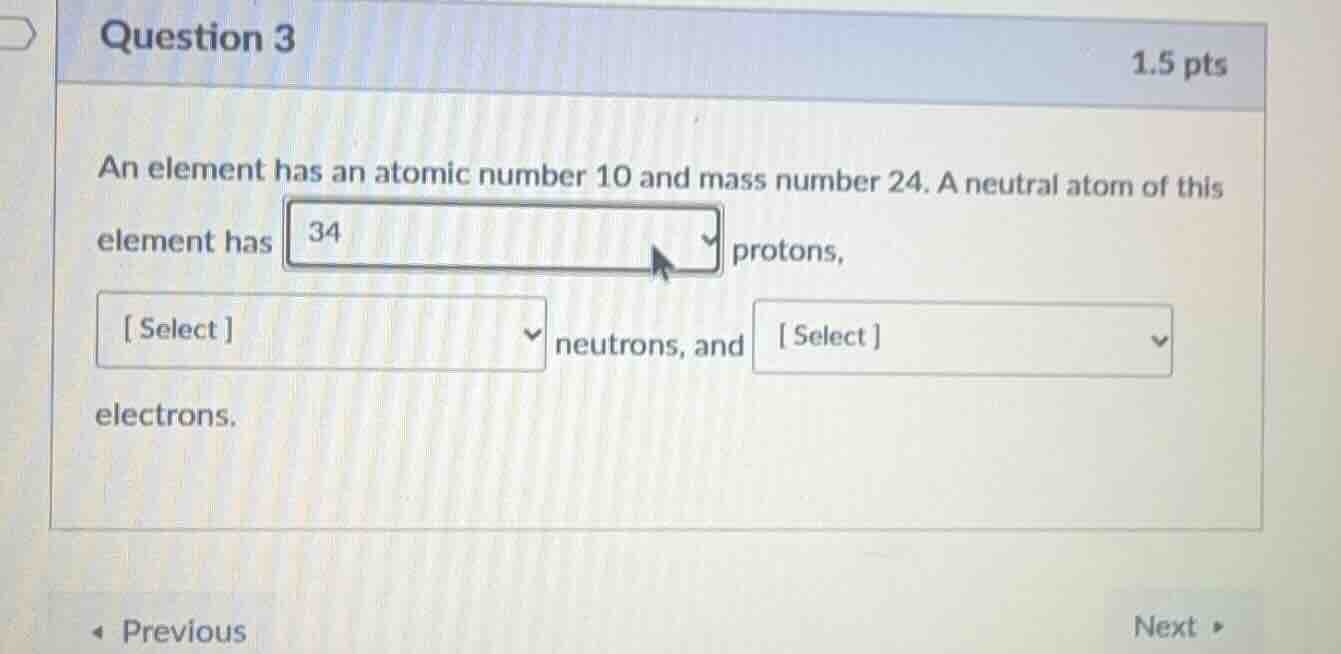
an element has an atomic number 10 and mass number 24. a neutral atom of this element has 34 protons,
select neutrons, and select electrons.
Step1: Find number of protons
Atomic number = number of protons. Given atomic number = 10, so protons = 10.
Step2: Calculate number of neutrons
Mass number = protons + neutrons. Rearranged: neutrons = mass number - protons.
$\text{Neutrons} = 24 - 10 = 14$
Step3: Find number of electrons
Neutral atom has equal protons and electrons. So electrons = 10.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Protons: 10, Neutrons: 14, Electrons: 10