QUESTION IMAGE
Question
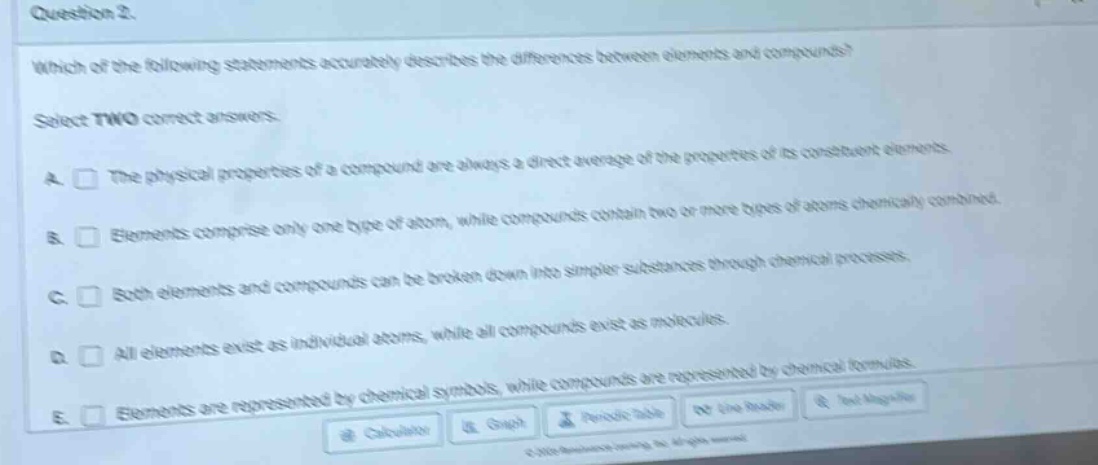
question 2.
which of the following statements accurately describes the differences between elements and compounds?
select two correct answers.
a. the physical properties of a compound are always a direct average of the properties of its constituent elements.
b. elements comprise only one type of atom, while compounds contain two or more types of atoms chemically combined.
c. both elements and compounds can be broken down into simpler substances through chemical processes.
d. all elements exist as individual atoms, while all compounds exist as molecules.
e. elements are represented by chemical symbols, while compounds are represented by chemical formulas.
- Option A: A compound's properties are not a direct average of its elements' properties (e.g., water vs. H and O). So A is incorrect.
- Option B: By definition, an element has one type of atom, and a compound has two or more types chemically bonded. This is correct.
- Option C: Elements cannot be broken down into simpler substances by chemical processes (they are the simplest form). So C is incorrect.
- Option D: Not all elements exist as individual atoms (e.g., diatomic elements like O₂), and not all compounds are molecules (e.g., ionic compounds like NaCl). So D is incorrect.
- Option E: Elements have chemical symbols (e.g., H for hydrogen), and compounds have chemical formulas (e.g., H₂O for water). This is correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. Elements comprise only one type of atom, while compounds contain two or more types of atoms chemically combined.
E. Elements are represented by chemical symbols, while compounds are represented by chemical formulas.