QUESTION IMAGE
Question
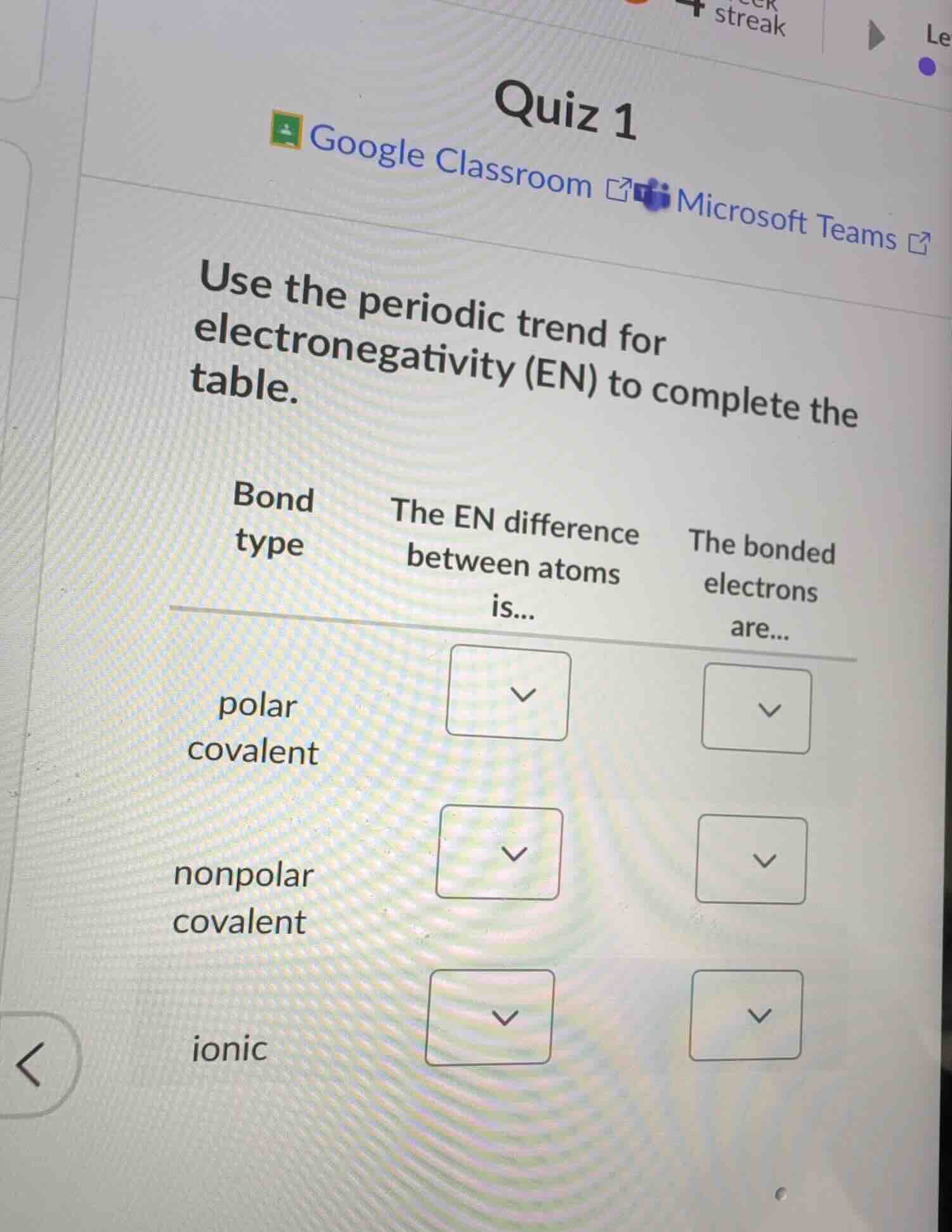
quiz 1
use the periodic trend for electronegativity (en) to complete the table.
bond type the en difference between atoms is... the bonded electrons are...
polar covalent
nonpolar covalent
ionic
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
polar covalent: The EN difference between atoms is... 0.4 to 1.7; The bonded electrons are... shared unequally
nonpolar covalent: The EN difference between atoms is... 0 to <0.4; The bonded electrons are... shared equally
ionic: The EN difference between atoms is... >1.7; The bonded electrons are... transferred (not shared, fully associated with one atom)