QUESTION IMAGE
Question
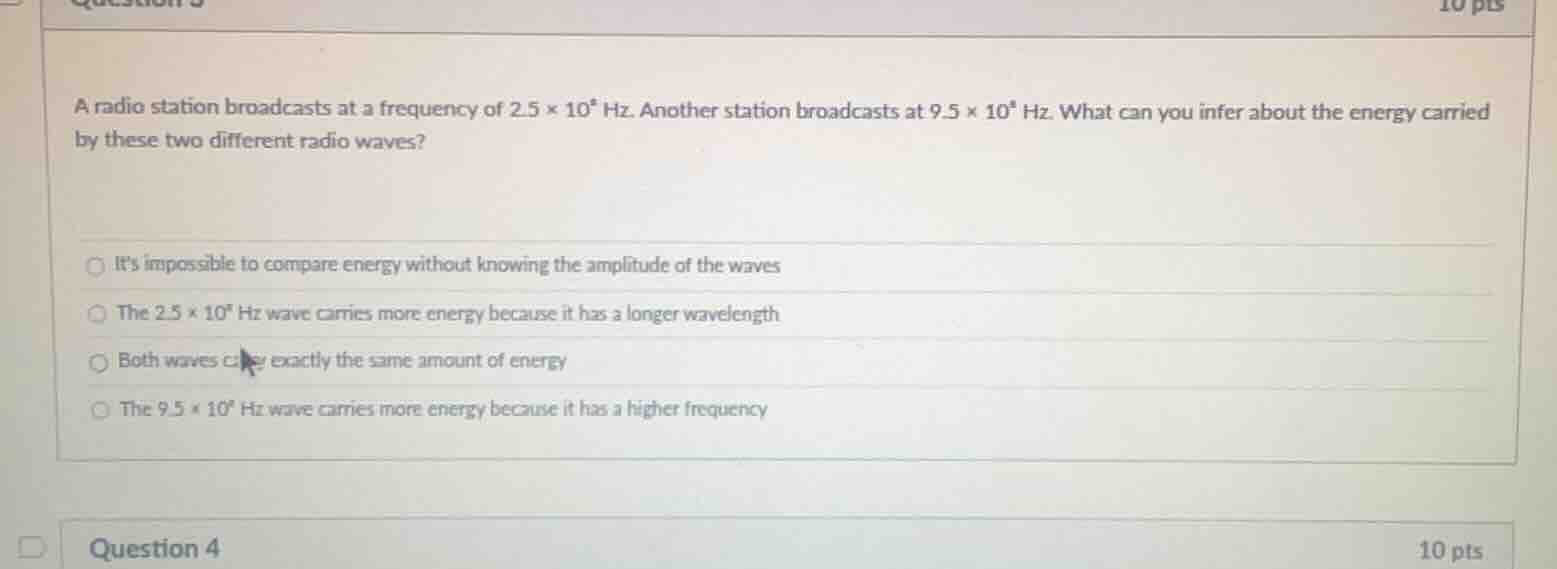
a radio station broadcasts at a frequency of $2.5 \times 10^8$ hz. another station broadcasts at $9.5 \times 10^8$ hz. what can you infer about the energy carried by these two different radio waves?\
\
\bigcirc its impossible to compare energy without knowing the amplitude of the waves\
\bigcirc the $2.5 \times 10^8$ hz wave carries more energy because it has a longer wavelength\
\bigcirc both waves carry exactly the same amount of energy\
\bigcirc the $9.5 \times 10^8$ hz wave carries more energy because it has a higher frequency
To determine the energy of a photon (and by extension, electromagnetic waves like radio waves), we use the formula \( E = hf \), where \( E \) is energy, \( h \) is Planck's constant, and \( f \) is frequency. Since \( h \) is a constant, energy is directly proportional to frequency. The first station has a frequency of \( 2.5 \times 10^{4}\, \text{Hz} \), and the second has \( 9.5 \times 10^{4}\, \text{Hz} \). The second station's frequency is higher, so its wave carries more energy.
- The first option is incorrect because the energy of a photon (for electromagnetic waves) depends on frequency (via \( E = hf \)) and amplitude is related to intensity (number of photons), not individual photon energy.
- The second option is incorrect because longer wavelength means lower frequency (since \( c = \lambda f \), \( c \) is constant), and lower frequency means lower energy (from \( E = hf \)).
- The third option is incorrect because the frequencies are different, so energies (per photon) are different.
- The fourth option is correct as higher frequency ( \( 9.5 \times 10^{4}\, \text{Hz} \) is higher than \( 2.5 \times 10^{4}\, \text{Hz} \)) means more energy per photon (from \( E = hf \)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. The \( 9.5 \times 10^{4}\, \text{Hz} \) wave carries more energy because it has a higher frequency