QUESTION IMAGE
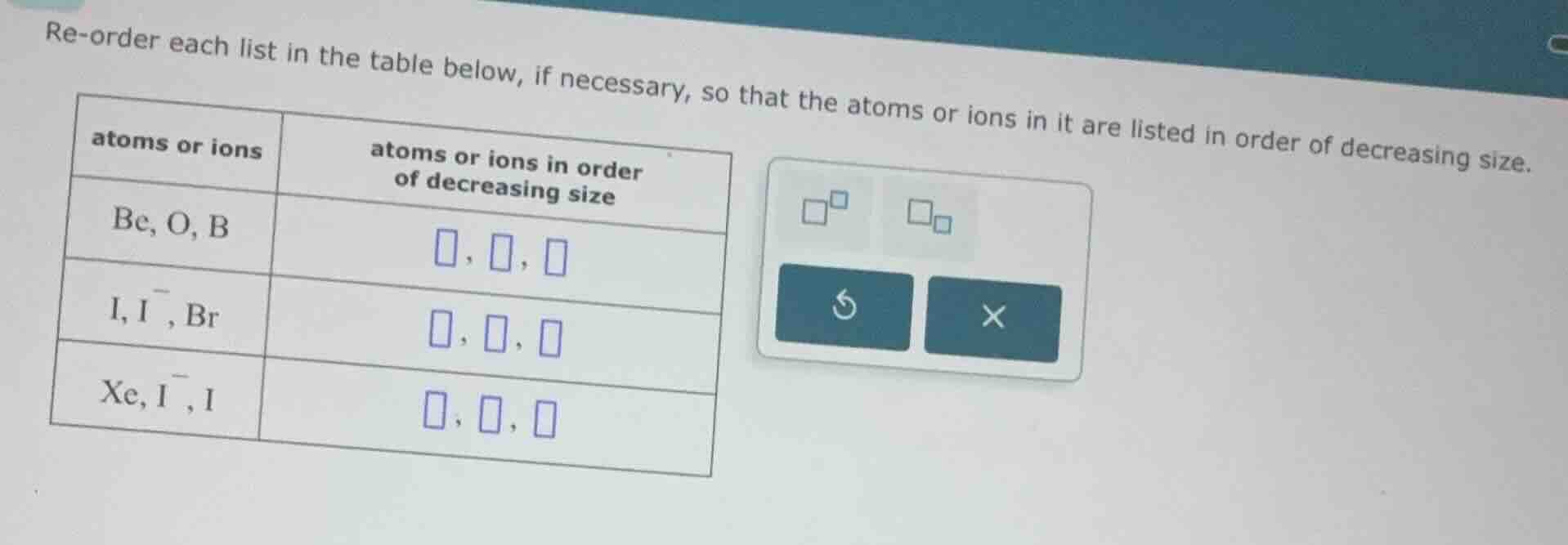
Question
re-order each list in the table below, if necessary, so that the atoms or ions in it are listed in order of decreasing size.
| atoms or ions | atoms or ions in order of decreasing size |
|---|---|
| i, i⁻, br | □, □, □ |
| xe, i⁻, i | □, □, □ |
Step1: Analyze period trend (Be, O, B)
Atomic size decreases across a period (left to right). Be (Group 2, Period 2) > B (Group 13, Period 2) > O (Group 16, Period 2).
Step2: Analyze group/ion trend (I, I⁻, Br)
Anions are larger than parent atoms; size increases down a group. I⁻ > I (Period 5) > Br (Period 4).
Step3: Analyze isoelectronic/group trend (Xe, I⁻, I)
I⁻ is isoelectronic with Xe; Xe (Period 5, noble gas) has larger radius than neutral I; anion I⁻ is larger than Xe. I⁻ > Xe > I.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Be, B, O
- I⁻, I, Br
- I⁻, Xe, I