QUESTION IMAGE
Question
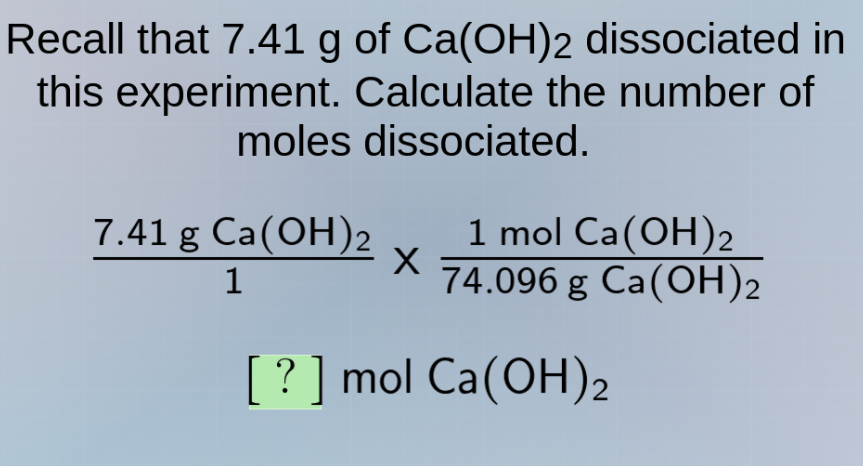
recall that 7.41 g of ca(oh)₂ dissociated in this experiment. calculate the number of moles dissociated. \\(\frac{7.41\text{ g }\ce{ca(oh)2}}{1}\times\frac{1\text{ mol }\ce{ca(oh)2}}{74.096\text{ g }\ce{ca(oh)2}}\\) \\(?\text{ mol }\ce{ca(oh)2}\\)
Step1: Multiply the given mass by the molar mass conversion factor
We have the mass of \( \text{Ca(OH)}_2 \) as \( 7.41 \, \text{g} \) and the conversion factor \( \frac{1 \, \text{mol}}{74.096 \, \text{g}} \). So we calculate \( 7.41 \times \frac{1}{74.096} \).
\( 7.41 \div 74.096 \approx 0.100 \) (rounded to three significant figures)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 0.100 \) (or approximately \( 0.10 \) depending on significant figure requirements, but the calculation gives approximately \( 0.100 \) when considering the given values)