QUESTION IMAGE
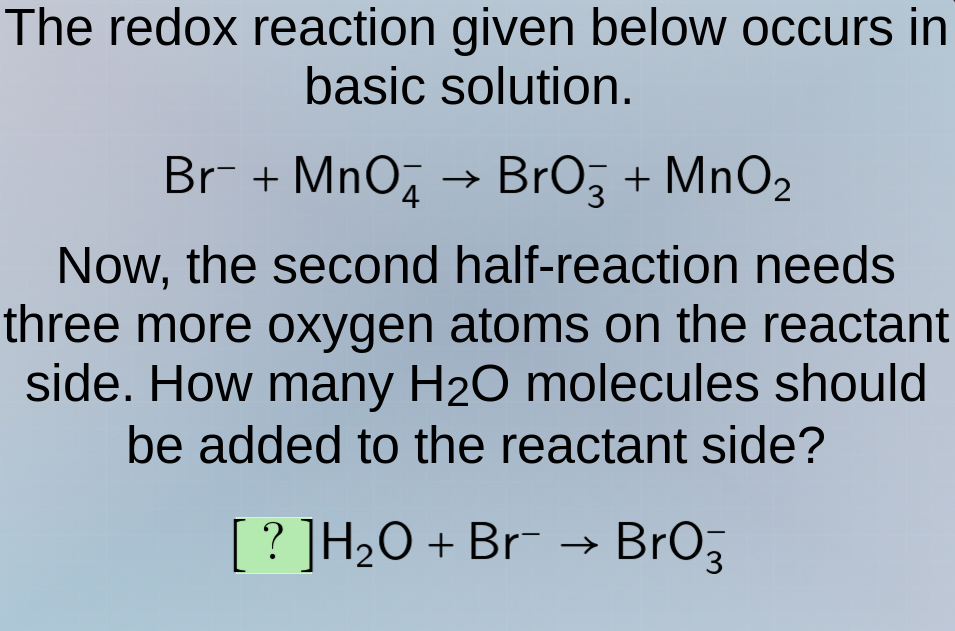
Question
the redox reaction given below occurs in basic solution.
br⁻ + mno₄⁻ → bro₃⁻ + mno₂
now, the second half - reaction needs three more oxygen atoms on the reactant side. how many h₂o molecules should be added to the reactant side?
?h₂o + br⁻ → bro₃⁻
Step1: Analyze oxygen atoms in H₂O
Each \( \ce{H_2O} \) molecule provides 1 oxygen atom.
Step2: Determine number of H₂O
We need 3 more oxygen atoms on the reactant side. Since each \( \ce{H_2O} \) gives 1 O, the number of \( \ce{H_2O} \) molecules needed is equal to the number of oxygen atoms needed. So, we need 3 \( \ce{H_2O} \) molecules.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
3